| Journal of Medical Cases, ISSN 1923-4155 print, 1923-4163 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Med Cases and Elmer Press Inc |
| Journal website http://www.journalmc.org |
Case Report
Volume 9, Number 8, August 2018, pages 278-280
Trans-Catheter Aortic Valve Replacement Thrombus in Patient on Xarelto and Aspirin Resolved Three Months After Warfarin
Obiora Maluduma, c, Firas Ajamb, Anas Alrefaeeb, Hetavi Mahidab, Nene Ugoekea, Dawn Calderona, Renato Apolitoa
aCardiovascular Disease Section, Department of Internal Medicine, Jersey Shore University Medical Center, Neptune, NJ, USA
bDepartment of Internal Medicine, Jersey Shore University Medical Center, Neptune, NJ, USA
cCorresponding Author: Obiora Maludum, Cardiovascular Disease Section, Department of Internal Medicine, Jersey Shore University Medical Center, Neptune, NJ 07753, USA
Manuscript submitted May 7, 2018, accepted June 20, 2018
Short title: TAVR Thrombosis on Direct Oral Anticoagulant
doi: https://doi.org/10.14740/jmc3084w
| Abstract | ▴Top |
Trans-catheter aortic valve replacement has been gaining ground since it was performed in 2002. Indications for the procedure are also expanding and are expected to include patients with intermediate risk for perioperative mortality. There are no guidelines on the use of anticoagulation in patients who have undergone trans-catheter aortic valve replacement. We present an interesting case of an 82-year-old gentleman with past medical history of paroxysmal atrial fibrillation with CHA2DS2-VASc (a clinical prediction tool for estimating risk of stroke in patients with atrial fibrillation) score of 3, coronary artery disease, pernicious anemia, and severe symptomatic aortic stenosis who had trans-catheter aortic valve replacement. He was on Xarelto and aspirin, and still developed thrombus on the aortic valve many months after the procedure. The thrombus resolved after few months of anti-coagulation with Coumadin. This case shows that thrombus on the valve can still occur post-procedure even in patients who are on direct oral anticoagulant and aspirin. This calls into question if there is need to anti-coagulate all patients after TAVR procedure with Coumadin for at least 3 - 6 months. As our knowledge of the post-TAVR complications and outcomes continues to grow, we may be able to answer this question in the future, especially with large scale research studies.
Keywords: TAVR; Aortic prosthetic valve; Thrombosis; Anticoagulation
| Introduction | ▴Top |
The first trans-catheter aortic valve replacement (TAVR) procedure was performed in 2002 [1]. Since then, it has become the standard of care in prohibitive and high risk patients for surgical aortic valve replacement [2]. Since the first US Food and Drug Administration approval in 2011, over 50,000 patients have undergone TAVR in the United States alone [2]. Although clinical or symptomatic leaflet thrombosis is uncommon, it is a serious adverse event following TAVR with a grave prognosis.
As indications for TAVR is expected to expand in the future to include patients at intermediate and perhaps low risk for perioperative mortality following surgery, an early diagnosis of leaflet thrombosis and treatment with antithrombotic therapy would be critical to optimize clinical outcomes.
It was recorded that most cases appear to occur in the first year following TAVR, and the role of direct oral anticoagulants (DOAC) is being investigated in ongoing trials [3]. Accordingly, there is uncertainty on how to prevent and treat this complication in patients who present after 1 year while on DOAC.
| Case Report | ▴Top |
Our patient is an 82-year-old gentleman with a past medical history of paroxysmal atrial fibrillation with a CHA2DS2-VASc score of 3, coronary artery disease, pernicious anemia, and severe symptomatic aortic stenosis. Pre-procedural transthoracic echocardiography (TTE) showed an aortic valve area of 0.67 cm2 and preserved left ventricular function. A 29 mm Edwards Sapien III valve was successfully deployed without post dilatation. The post-procedural mean trans-valve gradient was 5 mm Hg, and no significant para-valvular regurgitation was observed by transesophageal echocardiogram (TEE). The patient opted for off-label Xarelto for anticoagulation because he was not doing well on warfarin, with very labile international normalized ratio (INR). A long discussion about the off-label use of Xarelto in the setting of bio-prosthetic aortic valve replacement led to his decision to pursue this along with aspirin for paroxysmal atrial fibrillation, stroke prophylaxis. He was then discharged on Xarelto 20 mg daily and aspirin 81 mg daily. He was otherwise doing well, and without dyspnea or chest tightness. On routine outpatient follow-up, we noted a murmur that was increased in intensity and repeat TTE showed that the gradients had increased to a mean gradient of 24 mm Hg. There was thickening of the aortic valve and thrombosis was concerning and suspected. There was no fever, chills, no rash, no weight loss, weight gain, or signs of endocarditis or infection, no tooth aches etc. The patient denied any nausea, vomiting, diarrhea. He did undergo TEE which confirmed right coronary cusp thrombus measuring about 1.43 cm by 0.91 cm, which was layered, non-mobile, with normal valve function except for mild restriction leaflets (Fig.1).
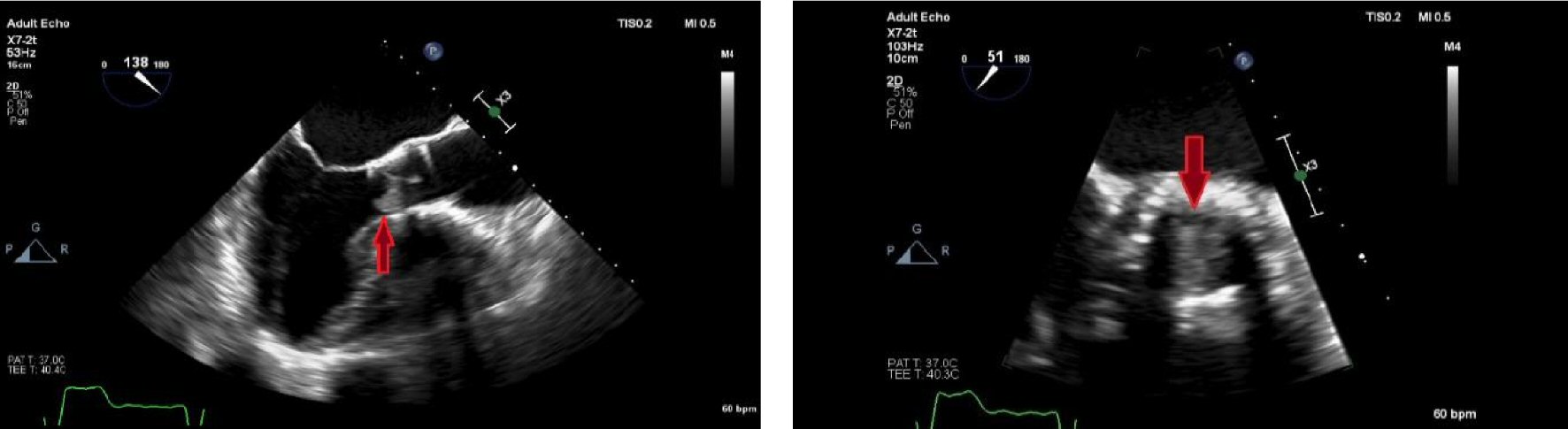 Click for large image | Figure 1. Echocardiographic pictures of THV before treatment. Red arrows point to the thrombus on THV in two different views. |
It was decided to admit him for high intensity IV heparin with bridge to warfarin and goal of INR between 2.5 and 3.5. He was discharged from the hospital few days later when his INR reached goal. Repeat TEE 3 months later showed complete resolution of the thrombus and normal valve function (Fig. 2).
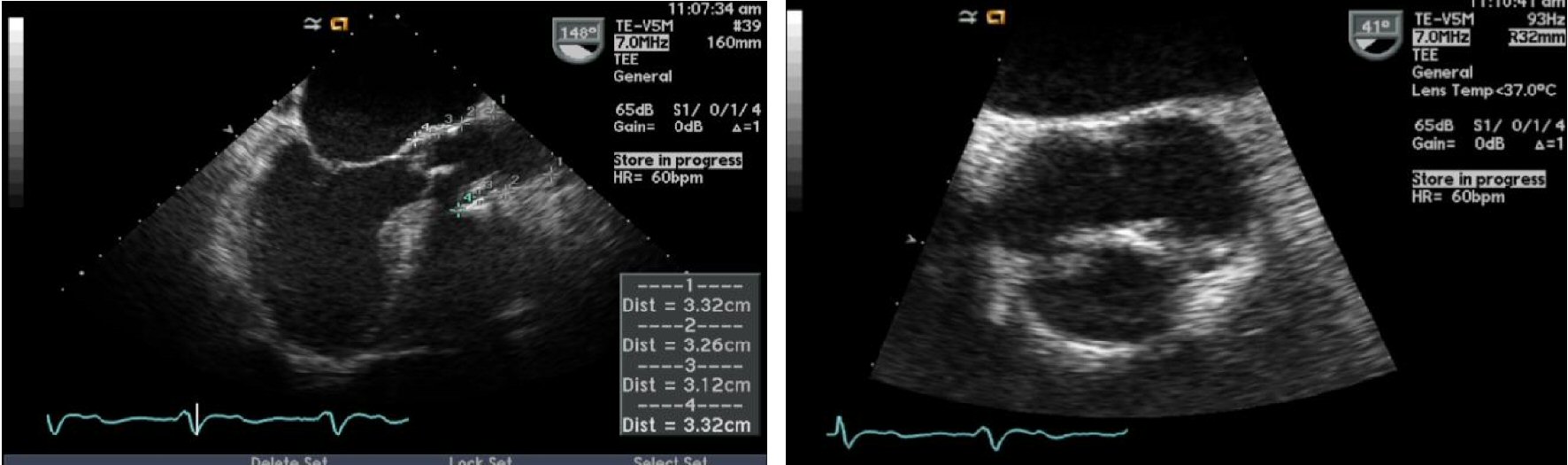 Click for large image | Figure 2. Echocardiographic pictures of the same patient showing resolution of the thrombus after treatment with warfarin. |
| Discussion | ▴Top |
In a recent large consecutive cohort of 405 patients having TAVR performed with the Edwards balloon-expandable trans-catheter heart valve (THV), the thrombus was diagnosed 1 - 3 months after the procedure by multi-detector computed tomography (MDCT) in 7% of the patients [4].
Based on previous studies, tt has been noticed that the occurrence of THV thrombosis was associated with large THV size and in cases in which warfarin was not used as an option for antithrombotic agent [4-8]. THV thrombosis is expected to occur in the early period after TAVR and has not previously been reported later than 7 months post-TAVR [4, 9]. This case showed that THV dysfunction or an increase in murmur intensity should evoke prompt investigation for possible thrombus formation.
The predisposing factors for TAVR thrombosis in our case were the presence of paroxysmal atrial fibrillation, the use of Xarelto instead of warfarin and the use of mono antiplatelet therapy.
In a recent meta-analysis that compared aspirin versus aspirin and clopidogrel following TAVR, there was no difference in the 30-day clinical and cerebrovascular adverse event rates; however, a trend toward more bleeding in the aspirin and clopidogrel group was demonstrated, also it was recorded that monotherapy with aspirin was associated with a THV thrombosis risk of 25%, and importantly, all patients who experienced obstructive THV thrombosis received antiplatelet therapy alone [10].
Our patient did not experience any kind of cardiovascular symptoms. The thrombosis was discovered on a regular follow-up at the office with his cardiologist, which stresses the importance of regular long-term follow-up and raises the question of how frequently and for how long should those patients receive echocardiographic assessment of their valves. Based on a study that was done among 460 consecutive patients who underwent TAVR with the Edwards Sapien XT or Sapien 3 (Edwards Lifesciences, Irvine, California), THV thrombosis occurred in 28 of 405 (7%) patients. A total of 23 patients had subclinical THV thrombosis, whereas five (18%) patients experienced clinically overt obstructive THV thrombosis [4].
There are no clear guidelines on how to treat THV thrombus or the choice of anticoagulant for prevention. We and others have observed incidence of THV thrombosis in patients who are not on warfarin, thus indicating that short-term warfarin treatment or even long-term NOAC anticoagulants treatment of THV thrombosis might not be sufficient in patients who are prone to developing THV thrombosis [6].
Conclusions
THV thrombosis may develop late after TAVR despite treatment with DOAC and aspirin, and can be reversed with warfarin. As our knowledge of the outcomes post-TAVR continues to increase, this may lead to development of guideline in the future about the use of anticoagulation, especially warfarin, and for how long following the implantation of the valve. Large scale randomized study may be needed in the future to guide the choice of anticoagulant and duration of treatment.
Conflict of Interest
No conflict of interest from any of the authors.
| References | ▴Top |
- Cribier A, Eltchaninoff H, Bash A, Borenstein N, Tron C, Bauer F, Derumeaux G, et al. Percutaneous transcatheter implantation of an aortic valve prosthesis for calcific aortic stenosis: first human case description. Circulation. 2002;106(24):3006-3008.
doi pubmed - Otto CM, Kumbhani DJ, Alexander KP, Calhoon JH, Desai MY, Kaul S, Lee JC, et al. 2017 ACC Expert consensus decision pathway for transcatheter aortic valve replacement in the management of adults with aortic stenosis: a report of the american college of cardiology task force on clinical expert consensus documents. J Am Coll Cardiol. 2017;69(10):1313-1346.
doi pubmed - Windecker S, Tijssen J, Giustino G, Guimaraes AH, Mehran R, Valgimigli M, Vranckx P, et al. Trial design: Rivaroxaban for the prevention of major cardiovascular events after transcatheter aortic valve replacement: Rationale and design of the GALILEO study. Am Heart J. 2017;184:81-87.
doi pubmed - Hansson NC, Grove EL, Andersen HR, Leipsic J, Mathiassen ON, Jensen JM, Jensen KT, et al. Transcatheter Aortic Valve Thrombosis: Incidence, Predisposing Factors, and Clinical Implications. J Am Coll Cardiol. 2016;68(19):2059-2069.
doi pubmed - Pache G, Schoechlin S, Blanke P, Dorfs S, Jander N, Arepalli CD, Gick M, et al. Early hypo-attenuated leaflet thickening in balloon-expandable transcatheter aortic heart valves. Eur Heart J. 2016;37(28):2263-2271.
doi pubmed - Makkar RR, Fontana G, Jilaihawi H, Chakravarty T, Kofoed KF, De Backer O, Asch FM, et al. Possible subclinical leaflet thrombosis in bioprosthetic aortic valves. N Engl J Med. 2015;373(21):2015-2024.
doi pubmed - Latib A, Naganuma T, Abdel-Wahab M, Danenberg H, Cota L, Barbanti M, Baumgartner H, et al. Treatment and clinical outcomes of transcatheter heart valve thrombosis. Circ Cardiovasc Interv. 2015;8(4):e001779.
doi - Mylotte D, Andalib A, Theriault-Lauzier P, Dorfmeister M, Girgis M, Alharbi W, Chetrit M, et al. Transcatheter heart valve failure: a systematic review. Eur Heart J. 2015;36(21):1306-1327.
doi pubmed - Pingpoh C, Pache G, Nawras D, Guenkel L, Sami K, Zeh W, Zimmer E, et al. Valve thrombosis 7 months after transcatheter aortic valve implantation. Ann Thorac Surg. 2014;98(3):1079-1081.
doi pubmed - Hassell ME, Hildick-Smith D, Durand E, Kikkert WJ, Wiegerinck EM, Stabile E, Ussia GP, et al. Antiplatelet therapy following transcatheter aortic valve implantation. Heart. 2015;101(14):1118-1125.
doi pubmed
This article is distributed under the terms of the Creative Commons Attribution Non-Commercial 4.0 International License, which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Journal of Medical Cases is published by Elmer Press Inc.


