| Journal of Medical Cases, ISSN 1923-4155 print, 1923-4163 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Med Cases and Elmer Press Inc |
| Journal website https://www.journalmc.org |
Case Report
Volume 12, Number 6, June 2021, pages 238-242
Large Vessel Stroke Following Multiple Other Strokes and Cardiomyopathy in a Forty-Nine-Year-Old COVID-19 Patient
Saira Chaughtaia, Rabail Soomroa, Khaula Chaughtaia, Waleed Anwaara, Zeeshan Chaughtaia, Arif Asifa, Mohammad A. Hossaina, b
aDepartment of Medicine, Jersey Shore University Medical Center, Hackensack Meridian Health, Neptune, NJ 07753, USA
bCorresponding Author: Mohammad A. Hossain, Internal Medicine Residency Program, Jersey Shore University Medical Center, Neptune, NJ 07753, USA
Manuscript submitted February 15, 2021, accepted March 2, 2021, published online March 24, 2021
Short title: Stroke and Cardiomyopathy in a COVID-19 Case
doi: https://doi.org/10.14740/jmc3682
| Abstract | ▴Top |
The novel coronavirus known as severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) has made its presence known on the centerstage of worldwide healthcare in 2020. Although it is widely known about its pulmonary presence and ensuing complications, evidence is emerging that there are other organ systems including the cardiovascular and cerebrovascular systems that may be damaged by this virus. There have been reports of large vessel stroke occurring in coronavirus disease 2019 (COVID-19) positive patients, with very few reported in the age group less than 50 years. In this case, we describe a previously healthy 49-year-old male who presented with signs of stroke, and was found to have the novel coronavirus as he had been suffering from upper respiratory tract symptoms for 3 weeks. He subsequently developed further large vessel stroke while in the hospital despite being started on antiplatelet therapy. He was also found to have new onset cardiomyopathy. He was started on anticoagulation and discharged with follow-up for cardiomyopathy testing outpatient. This case begs the question on which anticoagulation to utilize in COVID-19 positive patients to be effective in preventing thrombotic events. It is postulated that a pro-inflammatory state induced by the virus and the virus’ affinity for angiotensin converting enzyme-2 receptors in the cerebral vasculature are predispositions to cause a stroke. The virus also directly damages cardiac myocytes causing a number of cardiac complications including cardiomyopathy. It is crucial that guidelines on anticoagulation choice and indications for when to start anticoagulation be developed in order to prevent the more devastating consequences of thrombosis and embolism and their subsequent clinical sequelae.
Keywords: SARS-CoV-2; Cardiomyopathy; Stroke
| Introduction | ▴Top |
The rapidly evolving clinical scene of the coronavirus disease 2019 (COVID-19) pandemic is demonstrating to us new ways the virus is manifesting itself. The virus that causes COVID-19 has been named severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), a member of the coronavirus family [1]. It is widely known to cause pulmonary complications including respiratory failure. There is now emerging evidence that this infection is associated with thromboembolic complications, including stroke in a small percentage of patients affected [2-4]. Even more troubling is that this ischemic complication has been noted in a small number of patients less than 50 years of age. Another manifestation of the novel coronavirus now being identified is cardiomyopathy. We describe a case of a 49-year-old male with no medical problems other than a history of hypertension who presented to our emergency department with symptoms of a stroke and subsequently developed cardiomyopathy. He was tested for the novel coronavirus and sure enough, was found to be positive.
| Case Report | ▴Top |
A 49-year-old male presented to the emergency room with expressive aphasia and right facial droop noticed by his wife. His last known normal neurological status was 1 h prior to arrival to the hospital. He had also been experiencing cough, shortness of breath, diarrhea and abdominal pains for about 3 weeks. He had gone to a different emergency room 1 week ago and was diagnosed with an upper respiratory tract infection and was advised over the counter cough medicine after obtaining a chest X-ray that was normal. His past medical history included possible hypertension for which he was on medication at one point, but currently off medication. He is a non-smoker. His blood pressure was 131/86 mm Hg, heart rate was 120 beats per minute, temperature was 36.4 °C, respiratory rate of 15 breaths per minute, and pulse oxygenation was 99% on room air. His neurological examination was consistent with expressive aphasia with right sided facial weakness. His sensation and motor function was intact in all four extremities with no ataxia. His other physical examination was unremarkable. His lab work (Table 1) including complete blood count and comprehensive metabolic panel were normal except for a creatinine of 1.58 mg/dL (normal: 0.61 - 1.24 mg/dL). His international normalized ratio (INR) was 1.18 (normal: 0.88 - 1.15), partial thromboplastin time (PTT) was 27 s (normal: 26 - 39 s), D-dimer was 1,221 ng/mL (normal: < 500 ng/mL). His hemoglobin A1c was 5.8% (normal: < 5.7%), total cholesterol was 191 mg/dL (normal: < 200 mg/dL), triglycerides were 242 mg/dL (normal: 0 - 150 mg/dL), high-density lipoprotein (HDL) was 25 mg/dL (normal: 39 - 79 mg/dL), and low-density lipoprotein (LDL) was 107 mg/dL (normal: < 100 mg/dL).
 Click to view | Table 1. Initial Laboratory Results of the Patient |
In the emergency room, he received 325 mg of aspirin, and was assessed by a code stroke team. He was not recommended tissue plasminogen activator (tpA) by the tele-neurologist as he had inconsistent symptoms. His chest X-ray was normal, carotid ultrasound showed no stenosis, and computed tomography of the head showed subtle area of diminished attenuation in the right cerebellar hemisphere and left frontoparietal lobe for which a subtle area of infarction could not be excluded. Magnetic resonance imaging (MRI) of the brain completed the following day revealed acute to subacute infarcts in the right cerebellum and left and right frontoparietal cortical regions. He was started on aspirin 81 mg daily and atorvastatin 40 mg daily.
He was also tested for COVID-19 and his test result came back with a positive SARS-CoV-2 PCR. He was started on azithromycin, zinc, and vitamin C. He had an echocardiogram of the heart which revealed an ejection fraction of 26-30% (normal: 55-60%) with abnormal left ventricular diastolic function, mildly increased left ventricular wall thickness, mild mitral regurgitation, and moderate global hypokinesis. A cardiologist evaluated him and deemed that he needed to have a transesophageal echocardiogram and left heart catheterization to determine the etiology of the newly diagnosed cardiomyopathy once he is recovered from his acute ischemic event and COVID-19.
On hospital day 4, he was found to have new global aphasia and left upper extremity weakness. He received a stat computed tomography of the brain (Fig. 1), which revealed a new right middle cerebral artery (MCA) infarction involving the right frontoparietal lobe and insular cortex. At that point, he was loaded with 600 mg of clopidogrel and transferred to the neuro-intensive care unit. It was decided that since he failed antiplatelet therapy, that the antiplatelet medications aspirin and clopidogrel would be stopped and full-dose anticoagulation was commenced with enoxaparin.
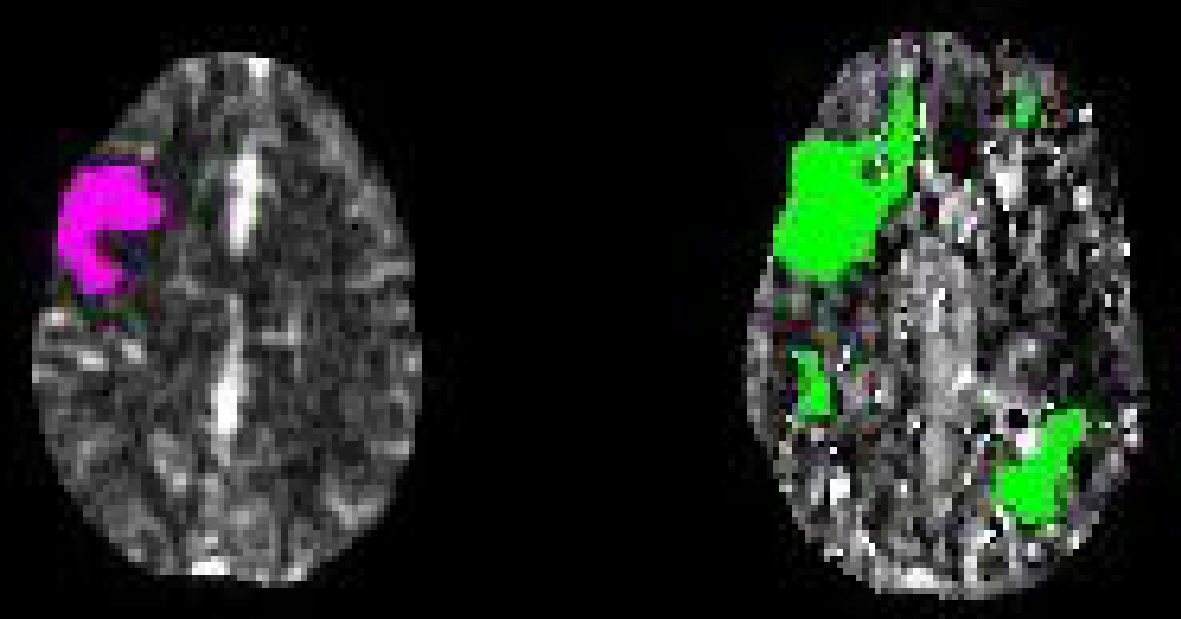 Click for large image | Figure 1. Computed tomography perfusion scan of head on hospital day 4 showing decreased perfusion within the cerebral hemispheres bilaterally (right image) appearing new within the right frontal region (left image) since the previous MRI. MRI: magnetic resonance imaging. |
He had hypercoagulability testing done, including antithrombin III, homocysteine, antinuclear antibody, beta-2-glycoprotein, cryoglobulin, and functional protein C, all of which were negative. He did have further testing of lupus anticoagulant which was positive; however, this test was not reliable as he had it done after being started on anticoagulation, which invalidates the reliability of the result.
For his cardiomyopathy, he was placed on a beta-blocker, angiotensin receptor blocker, and a diuretic. He had workup done for non-ischemic cardiomyopathy including human immunodeficiency virus (HIV), thyroid function tests, hepatitis serologies, and ferritin levels which were all within normal limits except for slightly elevated ferritin of 523 ng/mL (normal: 24 - 336 ng/mL), which was not high enough to cause cardiomyopathy. He remained in normal sinus rhythm throughout the hospital stay. He was advised to have repeat echocardiogram done on an outpatient basis along with ischemic workup including left heart catheterization.
He recovered from the symptoms of COVID-19 including shortness of breath and cough and on the day of discharge, he was saturating 98% on room air and was stable from a pulmonary standpoint.
His hospital course was further complicated by dysphagia requiring a gastric feeding tube, healthcare-associated pneumonia, and staphylococcus bacteremia requiring prolonged antibiotics. He was discharged on the novel oral anticoagulant apixaban and advised repeat hypercoagulability testing in 3 months. He went to a rehabilitation center.
| Discussion | ▴Top |
The novel coronavirus has rapidly transformed the landscape of the healthcare system in the year 2020. It has overshadowed and affected almost every aspect of the system from the outpatient world to the elective procedures world to the inpatient world. The virus is primarily known for its pulmonary involvement and its dreaded complications of respiratory failure and death [5]. What is now surfacing is the fact that the virus is now associated with ischemic strokes and cardiomyopathy in a small number of patients.
The virus has been shown to cause ischemic strokes in a small group of patients [1-5]. Whittaker et al did a review of neurologic complications identified to date and found a small study describing five patients identified with ischemic stroke in association with the novel coronavirus [1]. It is thought that these neurologic complications manifest themselves in patients with more severe disease [1]. This is unlike our patient who had mild symptoms of COVID-19 and whose respiratory status remained stable throughout hospitalization. Goldberg et al described a case of large vessel ischemic stroke arising in a patient who was previously diagnosed with SARS-CoV-2, and who required intubation for respiratory failure almost immediately upon arrival to the emergency department [2]. Worryingly, patients younger than 50 years of age have demonstrated to have this ischemic complication [2-4]. In a review of neurologic manifestations associated with COVID-19, there were three cases of ischemic stroke in which the patient was found with the stroke on imaging that was done for encephalopathy but without the typical sensory-motor symptoms of an ischemic event [3]. Again, neurologic manifestations were more commonly found in patients of severe COVID-19 illness [3]. Tunc et al described four cases in Turkey of COVID-19 patients found with either small or large vessel ischemic stroke, one patient of which was under the age of 50. They also had elevated D-dimer levels and C-reactive protein, both markers of inflammation [4]. There was also a similar case described of a patient with history of hypertension who presented with large vessel stroke and COVID-19, he was found to have elevated D- dimer levels and was eventually discharged with aspirin and statin medications [5]. In our patient’s case, he continued to have further strokes while on aspirin/statin therapy, and he was switched to full dose anticoagulation. Viguier et al described a case of acute cerebral ischemia precipitated by a common carotid artery thrombus [6]. This gives some evidence of hypercoagulable state in COVID-19 illness directly causing an acute ischemic event.
By now, it is established that the novel coronavirus infection is associated with thrombotic events. One study of 184 patients who were critically ill in the intensive care unit with COVID-19, found thrombotic events in 75 of them, five of whom with ischemic stroke [7]. It has been discovered that the virus has an affinity for angiotensin converting enzyme-2 (ACE2) receptors and attaches to them in the alveoli, as well as other locations including the brain vascular endothelial cells [8-10]. This in turn leads to the commencement of a pro-thrombotic state with elevated levels of D-dimer, fibrinogen, ferritin and other pro-inflammatory markers. Therefore, the virus may induce cerebral ischemia through a direct effect on ACE2 receptors and through a general pro-inflammatory state. A report from China of more than 200 patients pinpointed 6% of those patients to have an acute cerebral event [9]. Zhou et al commented that cerebral ischemia may occur up to 2 weeks after initial COVID-19 symptoms, similar in degree to our patient who presented 3 weeks after symptom onset. Patients may be treated with tPA or mechanical thrombectomy if they present within the window of time, so a high level of suspicion should be present for stroke in a patient presenting with stroke symptoms and evidence of possible SARS-CoV-2 infection [9, 10]. Providers caring for patients presenting with stroke during this pandemic should be keenly wary of any possible COVID-19 symptoms, and any suggestion of the virus should necessitate prompt testing and isolation for the virus [11]. The question remains for what the optimal antithrombotic therapy is to administer to patients of stroke induced by the novel coronavirus infection. The fact that our patient suffered another ischemic event after being started on antiplatelet therapy suggests that this is not sufficient. After being started on enoxaparin, the patient did not suffer any more ischemic events throughout his hospital stay. However, there have been reports of developing stroke in COVID-19 patients despite them being on full dose anticoagulation. Beyrouti et al described a patient with the novel coronavirus who was on full dose low-molecular-weight heparin for a pulmonary embolism, who subsequently developed posterior cerebral artery infarction [12]. Another patient who was taking warfarin for atrial fibrillation with an INR of 3.6 developed cerebral infarcts despite this [12]. There was a patient as young as 36 with COVID-19 who presented with cerebral infarction and ascending aortic thrombus [13]. It is alarming how thrombogenic the virus can be and therefore, we need proper guidelines on effective antithrombotic therapy and to initiate prophylactic versus therapeutic anticoagulation. Perhaps even more concerning, is the fact that patients are not presenting for stroke care as the numbers have dropped, this is likely due to the reluctance of patients to enter the healthcare system during this pandemic as to avoid exposure to the novel coronavirus [14]. An analysis study of 27,676 patients from 54 institutions found a prevalence of acute ischemic stroke of 1.3% of patients with COVID-19 versus 1.0% of patients without COVID-19 [15]. The patients with stroke and COVID-19 had much higher prevalence of risk factors like diabetes, hypertension, hyperlipidemia, and atrial fibrillation [15]. This is in contrast to our patient who was without risk factors and had a normal blood pressure on arrival despite possibly having hypertension in the past.
Also, of note, our patient had new onset cardiomyopathy with an ejection fraction of 26-30%. There have been few reports of different types of cardiomyopathy associated with COVID-19. One case described a 17-year-old male who died of cardiac arrest after having symptoms of COVID-19, post-mortem specimens identified eosinophilic myocarditis and he tested positive for SARS-CoV-2 [16]. There were also reported instances of takotsubo cardiomyopathy in association with COVID-19 [17-20]. There are a number of cardiac complications that may arise with severe and later disease stage including arrhythmias, heart failure, atrioventricular block and cardiomyopathy [21, 22]. There was even a report of two women developing cardiomyopathy in pregnancy associated with SARS-CoV-2 infection [23]. It is postulated that the ACE2 receptors are key in the viral damage that occurs to myocytes, along with a rise in inflammatory markers and cytokine damage [24-26].
It is possible that our patient’s stroke was a sequela of cardiomyopathy-induced cardio embolism. It is also possible that his stroke occurred by the mechanisms we know induce a hypercoagulable state in the novel coronavirus infection. Lastly, it is again possible that the stroke occurred from the direct toxic effects that the virus can have on cerebral vascular endothelium.
Conclusions
The novel coronavirus infection has drastically changed the landscape of healthcare in the year 2020. Aside from its known pulmonary manifestations, this virus has the potential to have toxic effects on other organ systems including the heart and brain. A deeper understanding of the array of complications associated with this infection will lead us to have a keener awareness and subsequently develop our knowledge of how to deal with these complications once they arise. Further guidelines need to be developed on which COVID-19 patients will require prophylactic anticoagulation therapy, to avoid the more devastating sequelae of cerebral infarction and all the issues that ensue from that.
Acknowledgments
None to declare.
Financial Declaration
This project was not supported by any grant or funding agencies.
Conflict of Interest
The authors declare that they have no competing interest.
Informed Consent
Written informed consent was obtained from the patient for publication of this case report and any accompanying images.
Author Contributions
SC contributed to the planning and case summary. RS, KC, WA, ZC all contributed to the discussion. MAH worked on the planning, manuscript revision and final approval. AA worked on the revision and final approval.
Data Availability
The authors declare that data supporting the findings of this study are available within the article.
| References | ▴Top |
- Whittaker A, Anson M, Harky A. Neurological Manifestations of COVID-19: A systematic review and current update. Acta Neurol Scand. 2020;142(1):14-22.
doi pubmed - Goldberg MF, Goldberg MF, Cerejo R, Tayal AH. Cerebrovascular Disease in COVID-19. AJNR Am J Neuroradiol. 2020;41(7):1170-1172.
doi pubmed - Ahmad I, Rathore FA. Neurological manifestations and complications of COVID-19: A literature review. J Clin Neurosci. 2020;77:8-12.
doi pubmed - Tun CA, UnlUba SY, Alemdar M, AkyUz E. Coexistence of COVID-19 and acute ischemic stroke report of four cases. J Clin Neurosci. 2020;77:227-229.
doi pubmed - Valderrama EV, Humbert K, Lord A, Frontera J, Yaghi S. Severe Acute Respiratory Syndrome Coronavirus 2 Infection and Ischemic Stroke. Stroke. 2020;51(7):e124-e127.
doi pubmed - Viguier A, Delamarre L, Duplantier J, Olivot JM, Bonneville F. Acute ischemic stroke complicating common carotid artery thrombosis during a severe COVID-19 infection. J Neuroradiol. 2020;47(5):393-394.
doi pubmed - Klok FA, Kruip M, van der Meer NJM, Arbous MS, Gommers D, Kant KM, Kaptein FHJ, et al. Confirmation of the high cumulative incidence of thrombotic complications in critically ill ICU patients with COVID-19: An updated analysis. Thromb Res. 2020;191:148-150.
doi pubmed - Hess DC, Eldahshan W, Rutkowski E. COVID-19-Related Stroke. Transl Stroke Res. 2020;11(3):322-325.
doi pubmed - Zhou Y, Li W, Wang D, Mao L, Jin H, Li Y, Hong C, et al. Clinical time course of COVID-19, its neurological manifestation and some thoughts on its management. Stroke Vasc Neurol. 2020;5(2):177-179.
doi pubmed - Qureshi AI, Abd-Allah F, Al-Senani F, Aytac E, Borhani-Haghighi A, Ciccone A, Gomez CR, et al. Management of acute ischemic stroke in patients with COVID-19 infection: Report of an international panel. Int J Stroke. 2020;15(5):540-554.
doi pubmed - Avula A, Nalleballe K, Narula N, Sapozhnikov S, Dandu V, Toom S, Glaser A, et al. COVID-19 presenting as stroke. Brain Behav Immun. 2020;87:115-119.
doi pubmed - Beyrouti R, Adams ME, Benjamin L, Cohen H, Farmer SF, Goh YY, Humphries F, et al. Characteristics of ischaemic stroke associated with COVID-19. J Neurol Neurosurg Psychiatry. 2020;91(8):889-891.
doi pubmed - Gonzalez-Pinto T, Luna-Rodriguez A, Moreno-Estebanez A, Agirre-Beitia G, Rodriguez-Antiguedad A, Ruiz-Lopez M. Emergency room neurology in times of COVID-19: malignant ischaemic stroke and SARS-CoV-2 infection. Eur J Neurol. 2020;27(9):e35-e36.
doi pubmed - Markus HS, Brainin M. COVID-19 and stroke-A global World Stroke Organization perspective. Int J Stroke. 2020;15(4):361-364.
doi pubmed - Qureshi AI, Baskett WI, Huang W, Shyu D, Myers D, Raju M, Lobanova I, et al. Acute Ischemic Stroke and COVID-19: An Analysis of 27 676 Patients. Stroke. 2021;52(3):905-912.
doi pubmed - Craver R, Huber S, Sandomirsky M, McKenna D, Schieffelin J, Finger L. Fatal Eosinophilic Myocarditis in a Healthy 17-Year-Old Male with Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2c). Fetal Pediatr Pathol. 2020;39(3):263-268.
doi pubmed - Roca E, Lombardi C, Campana M, Vivaldi O, Bigni B, Bertozzi B, Passalacqua G. Takotsubo syndrome associated with COVID-19. Eur J Case Rep Intern Med. 2020;7(5):001665.
doi pubmed - Nguyen D, Nguyen T, De Bels D, Castro Rodriguez J. A case of Takotsubo cardiomyopathy with COVID 19. Eur Heart J Cardiovasc Imaging. 2020;21(9):1052.
doi pubmed - Chadha S, Lodha A, Shetty V, Sadiq A, Hollander G, Frankel R, Shani J. Troponins in Tako-tsubo cardiomyopathy. Heart Lung Circ. 2013;22(9):775-777.
doi pubmed - Minhas AS, Scheel P, Garibaldi B, Liu G, Horton M, Jennings M, Jones SR, et al. Takotsubo Syndrome in the Setting of COVID-19. JACC Case Rep. 2020;2(9):1321-1325.
doi pubmed - Seecheran R, Narayansingh R, Giddings S, Rampaul M, Furlonge K, Abdool K, Bhagwandass N, et al. Atrial Arrhythmias in a Patient Presenting With Coronavirus Disease-2019 (COVID-19) Infection. J Investig Med High Impact Case Rep. 2020;8:2324709620925571.
doi pubmed - Kir D, Mohan C, Sancassani R. Heart brake: an unusual cardiac manifestation of COVID-19. JACC Case Rep. 2020;2(9):1252-1255.
doi pubmed - Juusela A, Nazir M, Gimovsky M. Two cases of coronavirus 2019-related cardiomyopathy in pregnancy. Am J Obstet Gynecol MFM. 2020;2(2):100113.
doi pubmed - Babapoor-Farrokhran S, Gill D, Walker J, Rasekhi RT, Bozorgnia B, Amanullah A. Myocardial injury and COVID-19: possible mechanisms. Life Sci. 2020;253:117723.
doi pubmed - Hendren NS, Drazner MH, Bozkurt B, Cooper LT, Jr. Description and proposed management of the acute COVID-19 cardiovascular syndrome. Circulation. 2020;141(23):1903-1914.
doi pubmed - Kochi AN, Tagliari AP, Forleo GB, Fassini GM, Tondo C. Cardiac and arrhythmic complications in patients with COVID-19. J Cardiovasc Electrophysiol. 2020;31(5):1003-1008.
doi pubmed
This article is distributed under the terms of the Creative Commons Attribution Non-Commercial 4.0 International License, which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Journal of Medical Cases is published by Elmer Press Inc.


