| Journal of Medical Cases, ISSN 1923-4155 print, 1923-4163 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Med Cases and Elmer Press Inc |
| Journal website https://www.journalmc.org |
Case Report
Volume 13, Number 9, September 2022, pages 462-470
Acquired Immune Deficiency Syndrome Cholangiopathy: Case Series of Three Patients and Literature Review
Yasir Ahmeda, f, Mustafeez Ur Rahmanb, Zoia Ehsan Khattakc, Jorge Herrerad, Eduardo Calderone
aDepartment of Internal Medicine, United Health Services Hospital (UHS), Binghamton, NY, USA
bPGY-III Internal Medicine at University of South Alabama Health University Hospital, Mobile, AL, USA
cKhyber Teaching Hospital, Peshawar, Pakistan
dSection of Hepatology, Department of Internal Medicine, USA Health, Mobile, AL, USA
eDepartment of Internal Medicine and Infectious Diseases, University of South Alabama Health University Hospital, Mobile, AL, USA
fCorresponding Author: Yasir Ahmed, Department of Internal Medicine, United Health Services Hospital (UHS), Binghamton, NY, USA
Manuscript submitted August 18, 2022, accepted September 22, 2022, published online September 28, 2022
Short title: AIDS Cholangiopathy
doi: https://doi.org/10.14740/jmc3998
| Abstract | ▴Top |
Cholangiopathy in acquired immune deficiency syndrome (AIDS) is being less frequently reported since antiretroviral therapy (ART) is available. It is associated with an advanced disease and seen in situations with poor access or non-compliance with ART. Liver biopsy is thought to have low yield in cases of AIDS cholangiopathy, but it can be an important tool in diagnosis, especially early in the course of the disease. The prognosis of AIDS cholangiopathy is generally not favorable, the therapy for opportunistic infections is mostly ineffective and restoration of immune system with ART remains the therapy of choice. We are sharing our experience of diagnosing and managing three cases of AIDS cholangiopathy.
Keywords: AIDS cholangiopathy; HIV/AIDS; Opportunistic infections; Antiretroviral therapy
| Introduction | ▴Top |
The cholangiopathy seen in patients with acquired immune deficiency syndrome (AIDS) is believed to have an infectious etiology, has characteristic cholangiographic findings, and is associated with advanced AIDS and low CD4 count [1-5]. Biliary tract abnormalities in AIDS patients were initially reported in the 1980s [6, 7]. AIDS cholangiopathy is commonly reported in young men who have sex with men with a mean age of 37 years [8, 9], and in heterosexual men in developing countries [10]. The true incidence of AIDS cholangiopathy is unknown and its incidence has decreased significantly since the initiation of antiretroviral therapy (ART) in mid-1990s [11]. Earlier studies estimated AIDS cholangiopathy to occur in 26% of patients with AIDS [11, 12] and one popular study early in 1990s estimated the prevalence to be as high as 30% in patients with chronic AIDS-related diarrhea [12]. It is now primarily seen in instances of poor access to ART and medication non-compliance, particularly in those with extremely low CD4 counts and opportunistic infections, as well as those with drug-resistant human immunodeficiency virus (HIV) infection [13]. We present three cases with relatively unique presentation and diagnostic challenges, our experience of managing these patients and the outcome.
| Case Reports | ▴Top |
Case 1
Investigations
A 28-year-old female with HIV infection and AIDS, and asthma, presented to the emergency department with acute onset abdominal pain, more so in the right upper quadrant (RUQ), for 1 week and increasing abdominal swelling for 3 days. She endorsed associated symptoms of nausea, vomiting and subjective fever and chills. She was afebrile and vitally stable on presentation, and abdomen was soft, though mildly distended and was diffusely tender to palpation. Scattered ecchymosis was also noticed on bilateral lower extremities. She was not able to adhere to ART or opportunistic infection prophylaxis (OIP).
Diagnosis
Pertinent laboratory data are mentioned in Table 1.
 Click to view | Table 1. Laboratory Workup for Case 1 |
Rest of the laboratory data were within normal limit. Abdominal ultrasound (US) showed features suggestive of cirrhosis associated with hepatomegaly and ascites, but no evidence of intrahepatic biliary ductal dilation or evidence of stones in the bile duct. Computed tomography (CT) scan of the abdomen with contrast showed hepatosplenomegaly and portal vein enlargement suggestive of portal hypertension.
Ascitic fluid analysis showed no evidence of spontaneous bacterial peritonitis (SBP). Hepatitis serologies excluded acute or chronic viral hepatitis. To further define the liver disease, a trans-jugular biopsy of the liver was performed which showed portal inflammation, cholangitis, ductular reaction, mild lobular inflammation, moderate steatosis, pericellular fibrosis without any bridging (Figs. 1-3). Additional data revealed 19,200 copies/µL of cytomegalovirus (CMV) on quantitative serum polymerase chain reaction (PCR). These findings in the setting of the clinical presentation suggested AIDS-related cholangiopathy with CMV infection as the likely etiology.
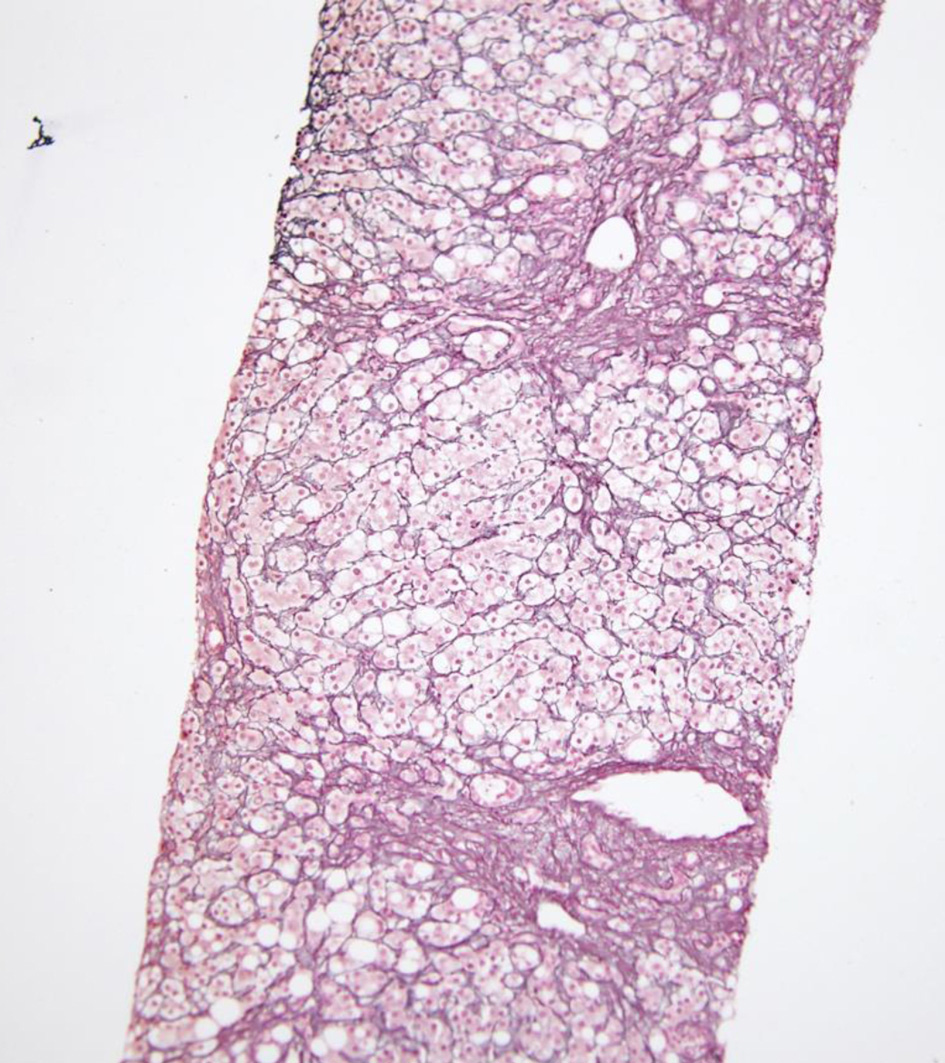 Click for large image | Figure 1. Preserved reticulin network with focal regenerative pattern on reticulin stain. |
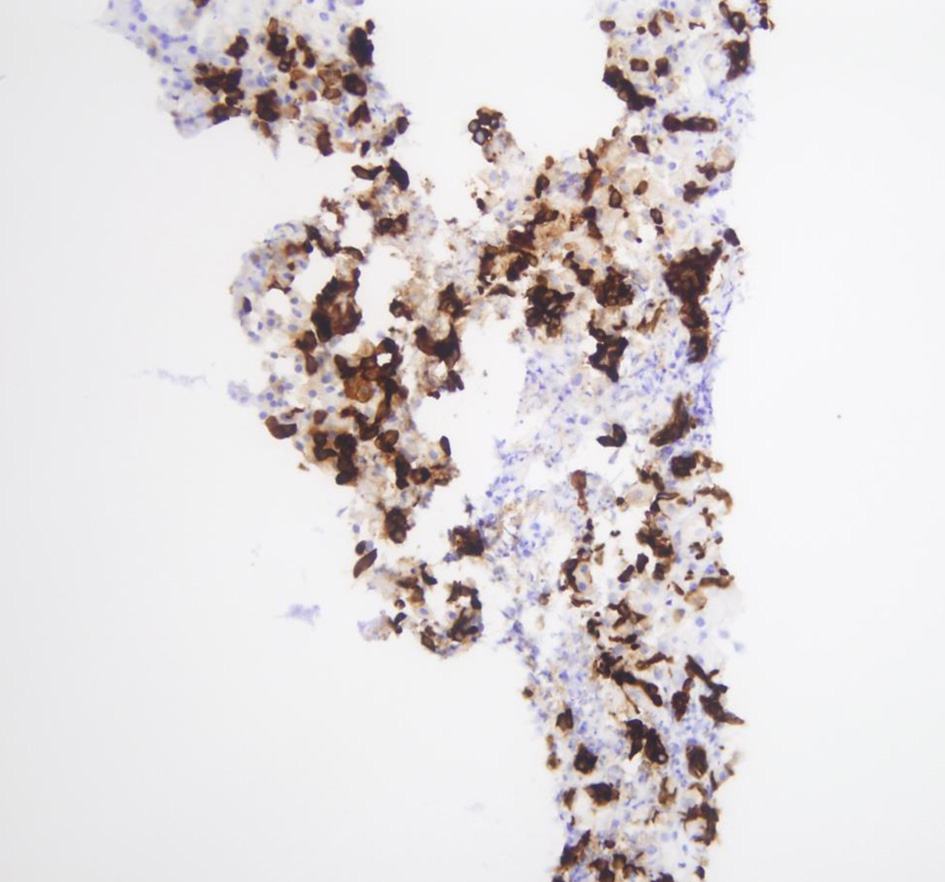 Click for large image | Figure 2. CK7 highlighting ductular reaction and scattered intermediate hepatocytes. |
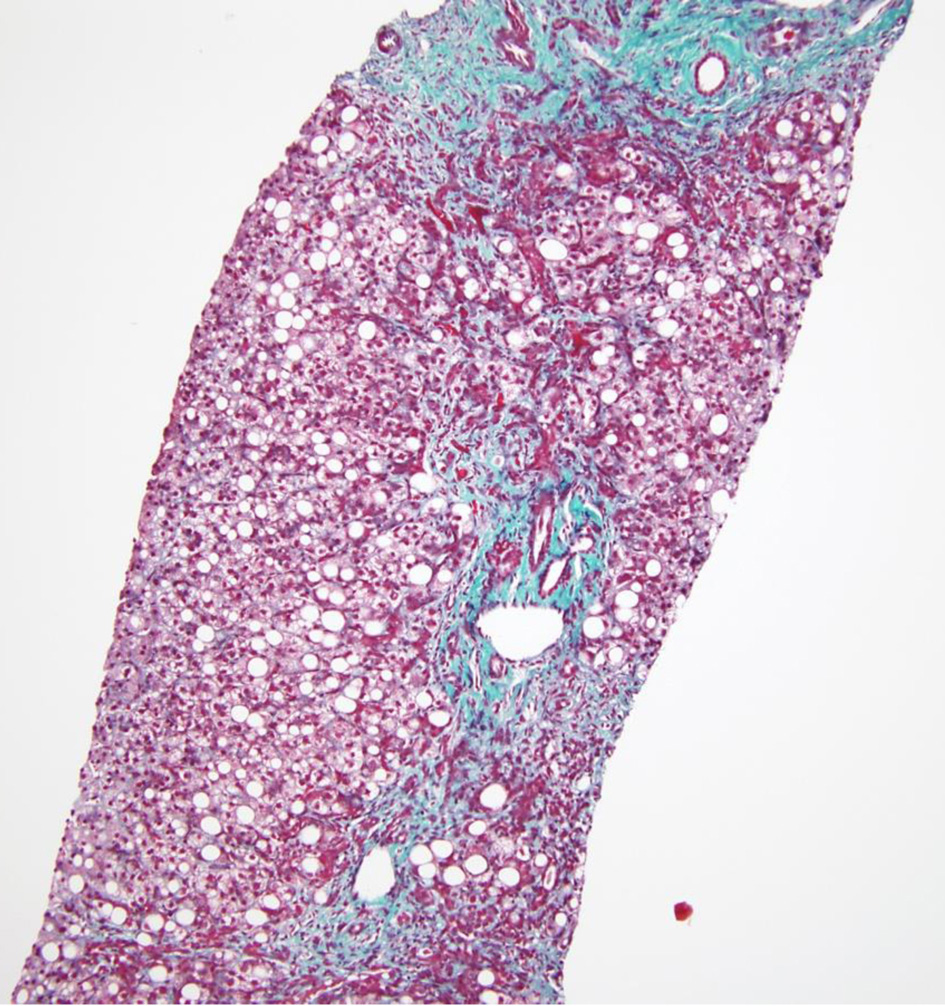 Click for large image | Figure 3. Trichome stain demonstrating pericellular and portal fibrosis without definite bridging. |
Treatment
Highly active retroviral therapy (HAART) was initiated, and the patient showed significant improvement in her symptoms.
Follow-up
The patient is doing well on her follow-up in the clinic 30 months after diagnosis.
Case 2
Investigations
A 38-year-old male with HIV infection, diabetes mellitus type II, and hypertension was transferred from a group home with failure to thrive for 2 months, and lower extremity edema and jaundice for 2 weeks. He also reported diarrhea for over 6 weeks. These symptoms were associated with night sweats, itching, loss of appetite and more than 20 pounds weight loss. On examination, he was vitally stable, had scleral icterus, severe cachexia with temporal wasting and bilateral lower extremity pitting edema. The abdomen was slightly tender to palpation in the RUQ and epigastric region. He reported not being able to take ART and engagement in sexual activity with men including receptive anal intercourse.
Diagnosis
Pertinent laboratory data are mentioned in Table 2.
 Click to view | Table 2. Laboratory Workup for Case 2 |
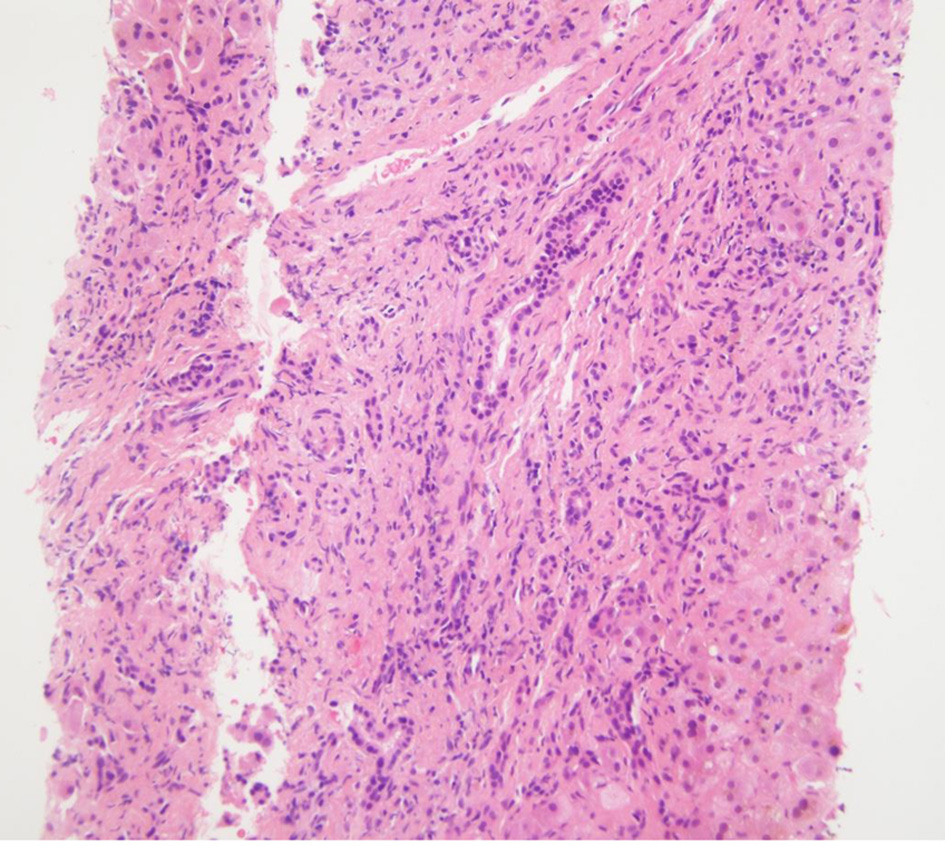
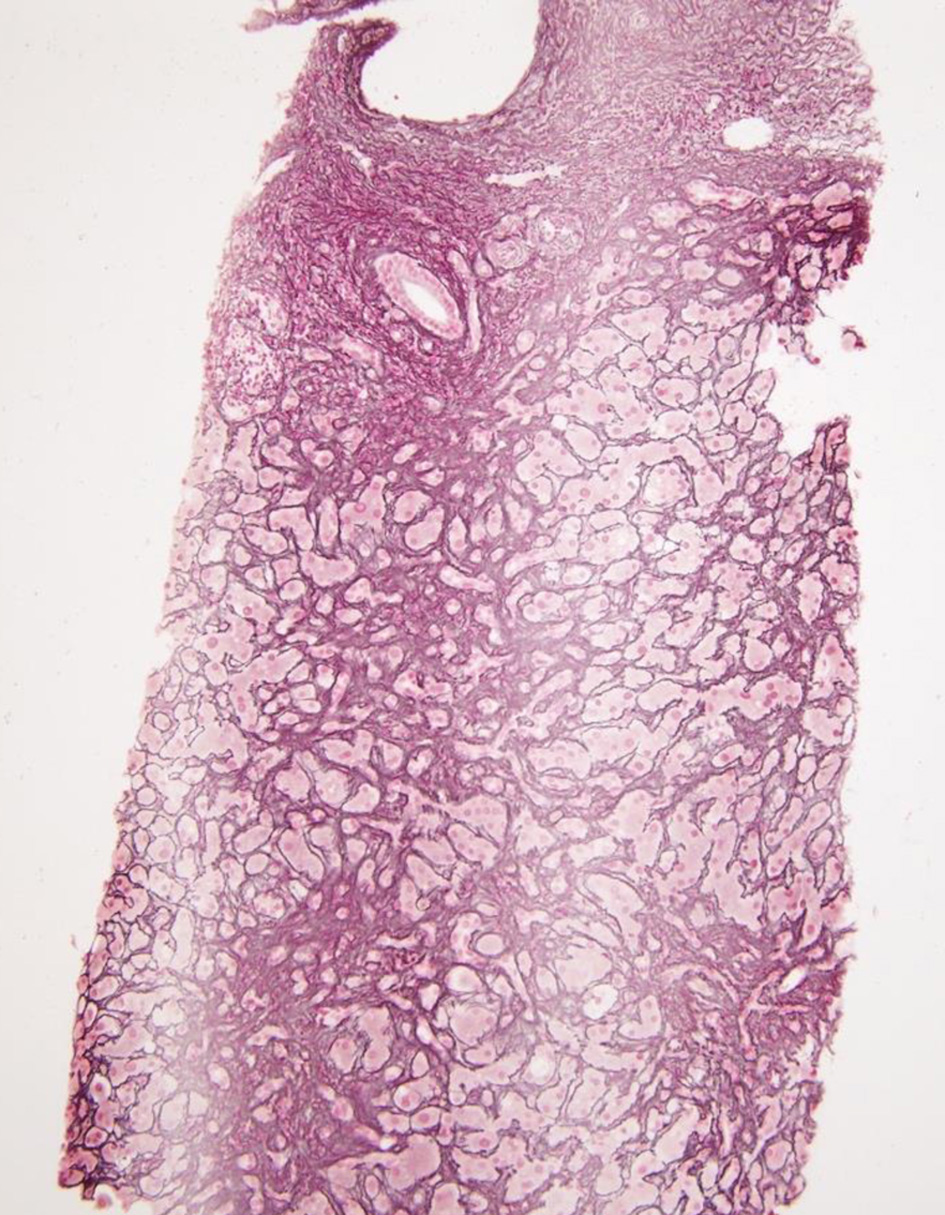
Rest of the liver profile was within normal limit. Stool pathogen testing panel was positive for microsporidium and quantitative serum PCR for CMV showed 6,910 copies/µL. An abdominal US showed hepatosplenomegaly with normal echogenicity, no evidence of intrahepatic biliary ductal dilatation, no evidence of stones and a small amount of free peritoneal fluid. Ascitic fluid analysis excluded SBP. Magnetic resonance cholangiopancreatography (MRCP) did not show evidence of pancreatic or biliary duct obstruction or cholelithiasis. A trans-jugular liver biopsy revealed cholestatic hepatitis with moderate to marked inflammation, prominent ductular proliferation and portal septal fibrosis with focal bridging (Figs. 4-6). These findings in the setting of AIDS and opportunistic infection with CMV and microsporidium were consistent with the diagnosis of AIDS-related cholangitis. CMV was not identified on rectal biopsy obtained via flexible sigmoidoscopy.
 Click for large image | Figure 4. Preserved reticulin network on reticulin stain. |
 Click for large image | Figure 5. Lympho-monocytic infiltrate admixed with plasma cells on H&E. H&E: hematoxylin and eosin. |
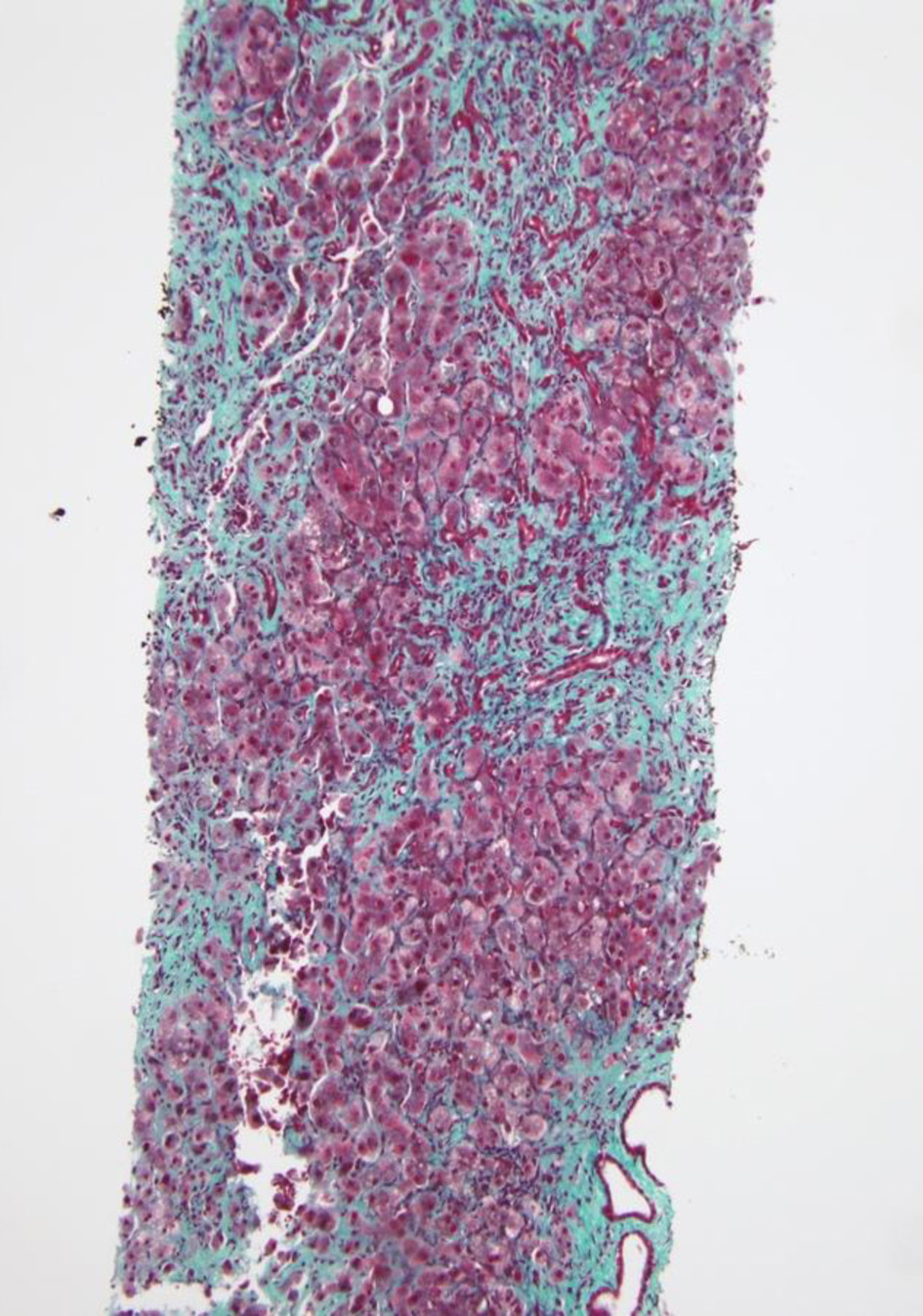 Click for large image | Figure 6. Portal tracts expansion by fibrosis with septa formation and focal bridging on trichome staining. |
Treatment and follow-up
Infectious disease team recommended albendazole and deemed that therapy for CMV was not warranted at that time. The patient expired within 4 weeks of diagnosis.
Case 3
Investigations
A 27-year-old male with asthma and hypertension visited the gastroenterology clinic for abdominal pain, diarrhea with intermittent blood streaking for 3 - 4 weeks. He also endorsed dysphagia. On physical examination, patient was vitally stable and had slight tenderness in the right upper and left lower quadrant. Digital rectal exam showed anal tags versus condylomas.
Diagnosis
Pertinent laboratory data are mentioned in Table 3.
 Click to view | Table 3. Laboratory Workup for Case 3 |
Total bilirubin was elevated and had a rising trend, with a value recorded as high as 15.2 mg/dL. Hepatitis serologies excluded acute or chronic viral hepatitis. HIV antibody testing was positive and a new diagnosis was made in this patient. HIV viral load was high and absolute CD4 count was low as shown in the table. Stool pathogen testing panel was positive for microsporidium. Quantitative serum PCR for CMV showed 9,750 copies/mL. Patient underwent flexible sigmoidoscopy and CMV was noted on immune-histochemical stain of rectal tissue biopsy, confirming the diagnosis of disseminated CMV infection.
Abdominal US was normal. MRCP did not show evidence of cholelithiasis or biliary system abnormalities either. An endoscopic retrograde cholangiopancreatography (ERCP) was done as bilirubin was elevated and right intrahepatic system images were missing on MRCP. It showed edematous and strictured common bile duct (CBD) (Fig. 7), from the distal to the proximal portion and its bifurcation. No filling defects were seen. The CBD and common hepatic duct (CHD) were narrowed preventing any contrast from being seen on cholangiogram (Fig. 8). These findings pointed towards a diagnosis of AIDS cholangiopathy.
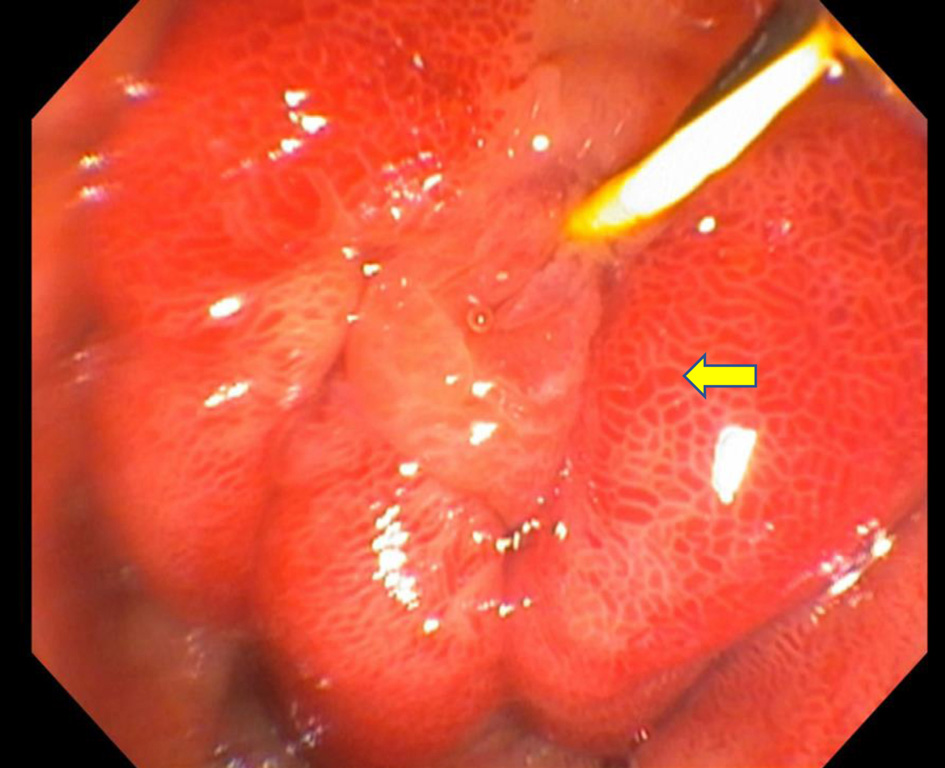 Click for large image | Figure 7. Major papilla among edematous mucosa and erythema. |
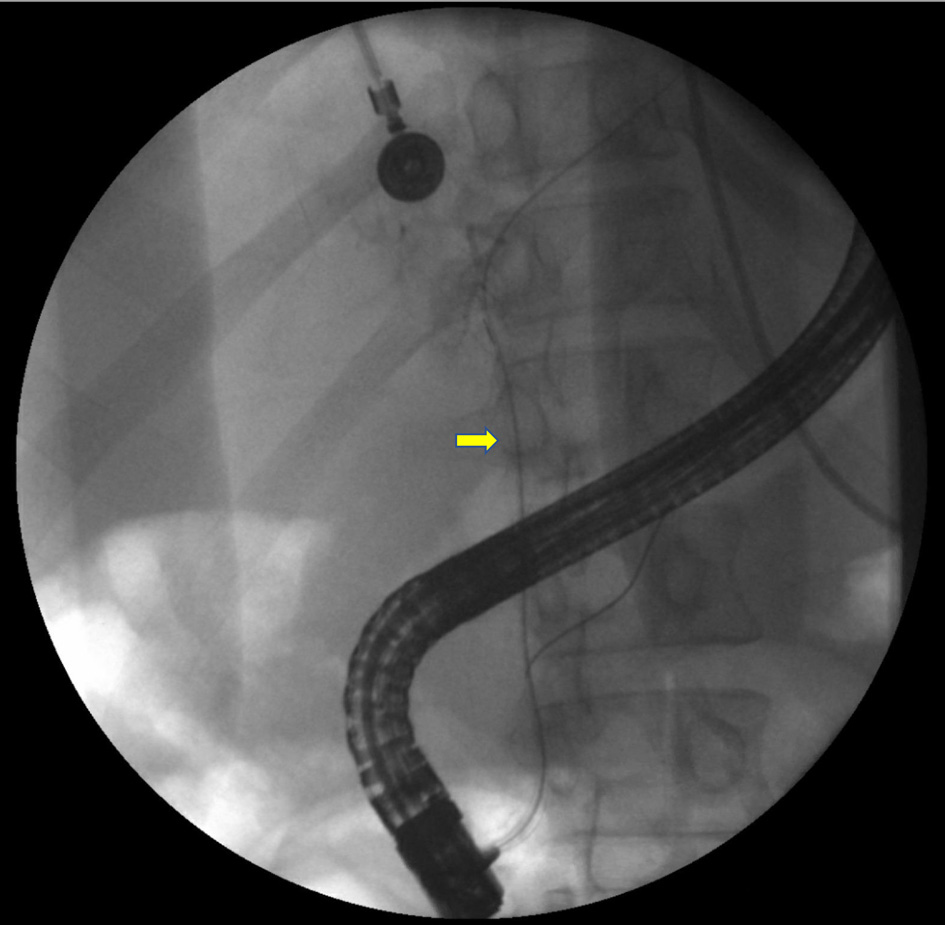 Click for large image | Figure 8. Minimal or no contrast on the film seen after maximal contrast injection. |
Treatment and follow-up
Albendazole and foscarnet were started on the recommendation of infectious disease team. Tenofovir and emtricitabine were initiated for the new diagnosis of HIV.
Patient had a prolonged hospital stay and continued to decline clinically. He expired within a month of diagnosis of AIDS cholangiopathy.
A summary of cases is shown in Table 4.
 Click to view | Table 4. Summary of Cases |
| Discussion | ▴Top |
AIDS cholangiopathy is thought to be caused by opportunistic infections in the biliary tract and in the setting of advanced immunosuppression [13]. The most common pathogen is Cryptosporidium parvum (C. parvum), isolated in up to 57% of patients [4]. In vitro studies suggest that C. parvum induces apoptotic cell death through Fas/Fas ligand (FasL) system and synergistic effects of the HIV-1 trans-activator of transcription (Tat) protein [14]. Another proposed mechanism is autonomic nerve damage caused by C. parvum, resulting in sphincter of Oddi dysfunction and papillary stenosis and CBD dilation [5]. Epidemiologic studies support the role of cryptosporidium as an etiologic agent of AIDS cholangiopathy [12]. CMV is the next-most common pathogen implicated in the pathogenesis of AIDS cholangiopathy, estimated to cause up to 20% of cases of AIDS cholangiopathy [13]. CMV damages the arterioles adjacent to the biliary canaliculi leading to ischemic damage in the biliary tree [1].
Microsporidia particularly Enterocytozoon bieneusi (E. bieneusi) and isospora have been found to be associated with AIDS cholangiopathy in a small percentage of patients, often in association with chronic diarrhea [13]. Cyclospora, giardia and Mycobacterium avium intracellulare (MAI) complex are very rare causes, contributing to less than 5% cases [1, 15, 16], while in some cases, no infectious pathogen is identified.
Clinically, AIDS cholangiopathy may be asymptomatic, but in most cases, RUQ pain (99%) is the most common presenting symptom, usually most severe in those with papillary stenosis [13]. Fever, nausea and vomiting, and jaundice are less common [3, 8, 10, 13]. Other symptoms may include diarrhea and symptoms of malabsorption, more commonly seen with microsporidium, cryptosporidium, or MAI infection [12, 17].
Serologic testing usually reveals profound immunosuppression with a low CD4 count. Liver tests are elevated in a cholestatic pattern as evidenced by marked increase in alkaline phosphatase (ALP) and gamma glutamyl transferase (GGT) levels in 80% of cases [3, 5, 8-13]. Transaminases are usually mildly elevated, and total bilirubin level is often normal or slightly elevated [5, 10, 13].
The diagnosis of AIDS cholangiopathy is usually established by imaging studies and/or liver biopsy. Abdominal US is the most common initial imaging modality [5]. Intra- or extrahepatic biliary dilation is the most common finding, but in early cases, it may be normal [9, 18, 19]. In two-thirds of the patients, an echogenic nodule at the distal end of CBD is noticed, which correlates with ampullary edema on ERCP [19].
In most cases, MRCP is performed to confirm or establish the diagnosis. One of the following four patterns is seen on cholangiography with MRCP or ERCP: 1) sclerosing cholangitis in combination with papillary stenosis (seen in 50% cases); 2) papillary stenosis alone; 3) combined intrahepatic and extrahepatic sclerosing cholangitis without papillary stenosis; and 4) long extrahepatic bile duct strictures with or without intrahepatic sclerosing cholangitis. MRCP is preferred over ERCP as it is non-invasive, does not have the risks of iatrogenic complications and is as sensitive as ERCP. The characteristic beaded appearance of sclerosing cholangitis is seen in only 20% of patients [1, 3, 9, 18-21]. Contrast-enhanced CT scan has a limited role in diagnosis and is most helpful in excluding other causes of cholestasis [22].
In the absence of typical cholangiographic findings, liver biopsy is often used to make the diagnosis. Findings similar to sclerosing cholangitis in non-AIDS patients are seen on liver biopsy in up to 50% in AIDS cholangiopathy [23]. Liver biopsy is most useful in early cases that have not developed the classical findings on cholangiography [24]. It can also exclude other potential causes of elevated liver tests and in some cases, help identify opportunistic organism causing cholestatic injury [25].
The management of AIDS cholangiopathy is mostly supportive and geared towards restoration of immune system. Treatment of opportunistic infections leading to AIDS cholangiopathy is rarely affective unless the immune system is restored. Nitazoxanide is the only US Food and Drug Administration (FDA) approved drug for cryptosporidium [26]. It is effective in 93% of non-AIDS patients [27], but the effectiveness is not replicated in AIDS patients [28]. Paromomycin is less effective alternative therapy for cryptosporidiosis in AIDS [17, 29], and azithromycin, spiramycin and bovine anti-cryptosporidium immunoglobulin were reported to be effective in some case series but were ineffective in controlled trials in AIDS patients and these trials were unfortunately never published [30].
CMV-directed treatment in AIDS cholangiopathy is rarely effective. Intravenous ganciclovir and foscarnet have shown no benefit [3, 9, 23, 31]. In microsporidia, transient therapeutic effect has been reported with albendazole [32]. There is some success reported with trimethoprim/sulfamethoxazole (TMP-SMX) for the treatment of Cyclospora in AIDS [33, 34], while it is recommended to combine TMP-SMX with ivermectin for isospora [35].
Ursodeoxycholic acid (UDSA) is a bile acid often used in the treatment of cholestatic liver disease. The experience in AIDS is very limited. Relief of abdominal pain and decreasing serum ALP was reported in four patients who were treated with URSO at 10 mg/kg/day [36]. No data on long-term benefit were provided.
ART appears to be the best treatment option for AIDS cholangiopathy that helps by restoring immune system, thus improving the probability of remaining free of AIDS and improved survival [37, 38]. It has also been shown to modify the course of disease and eradicate cryptosporidiosis and microsporidiosis in HIV-positive patients with diarrhea when a combination of ART is used [39, 40]. A combination ART including protease inhibitor resulted in restoration of immunity and clinical, microbiologic, and histologic response in eight out of nine HIV-infected patients with chronic gastrointestinal cryptosporidiosis, microsporidiosis, or both, while antimicrobial therapy with paromomycin, albendazole, and azithromycin was not effective in the same study [41]. In some patients, progression of AIDS cholangiopathy occurs despite successful ART therapy, suggesting that delay in diagnosis may lead to irreversible damage of the biliary system [42-44].
For patients with papillary stenosis, ERCP with sphincterotomy may provide a symptomatic relief. A statistically significant decrease in abdominal pain score after biliary sphincterotomy compared with a pre-procedure score was seen at 3.9 months follow-up in 12 out of 13 patients who underwent sphincterotomy during ERCP for papillary stenosis alone or in combination with sclerosing cholangitis [1]. In a study of 25 patients with AIDS associated with biliary stenosis who underwent ERCP sphincterotomy, patients were followed up long term (9.4 ± 1.2 months) and significant improvement was seen in abdominal pain score, while serum ALP remained essentially unchanged [2].
The prognosis of AIDS cholangiopathy is generally poor and is attributed to the presence of advanced AIDS. Median survival rates of 7 - 12 months in the pre-ART era have improved to 34 months. The presence of opportunistic infections and marked elevation of ALP appear to be associated with poor prognosis [45].
Conclusion
Since the introduction of HAART, the incidence of AIDS cholangiopathy has dramatically decreased. Individuals presenting with AIDS and elevated liver tests in a cholestatic pattern require aggressive evaluation to exclude AIDS cholangiopathy as delayed diagnosis results in irreversible biliary damage and poor prognosis. Liver biopsy is an important investigative tool in cases where classical findings on cholangiography are not present.
Learning points
Early recognition is important and AIDS cholangiopathy should be differentiated from other infectious diseases of the gastrointestinal tract.
Liver biopsy is an important diagnostic tool, especially earlier in the course of the disease.
Treatment with ART should be initiated in attempt to restore immune system.
Acknowledgments
None to declare.
Financial Disclosure
None to declare.
Conflict of Interest
None to declare.
Informed Consent
An consent was obtained from the patient for publication from one patient for publication of this case report and any accompanying images. We could not reach the next of kin for the other two patients who had expired.
Author Contributions
Yasir Ahmed contributed to the conceptualization, drafting the case series, introduction and case presentations, literature review, and final review and editing. Mustafeez Ur Rahman: contributed to the literature review in the preliminary draft, collecting pictures/slides and checking references. Zoia Ehsan Khattak contributed to the literature review in the preliminary draft. Jorge Herrera contributed to the review and editing of the draft and revision to improve suitability for publication. Eduardo Calderon, supervisor, contributed to the conceptualization, review and editing the draft and final review of the draft.
Data Availability
The authors declare that data supporting the findings of this study are available within the article.
Abbreviations
HIV: human immunodeficiency virus; AIDS: acquired immune deficiency syndrome; ART: antiretroviral therapy; HAART: highly active antiretroviral therapy; RUQ: right upper quadrant; OIP: opportunistic infection prophylaxis; ALT: alanine transaminase; AST: aspartate aminotransferase; ALP: alkaline phosphatase; GGT: gamma glutamyl transferase; SBP: spontaneous bacterial peritonitis; PCR: polymerase chain reaction; MRCP: magnetic resonance cholangiopancreatography; ERCP: endoscopic retrograde cholangiopancreatography; CBD: common bile duct; CHD: common hepatic duct; E. bieneusi: Enterocytozoon bieneusi; CMV: cytomegalovirus; C. parvum: Cryptosporidium parvum; FDA: Food and Drug Administration; TMP-SMZ: trimethoprim/sulfamethoxazole; UDSA: ursodeoxycholic acid
| References | ▴Top |
- Cello JP. Acquired immunodeficiency syndrome cholangiopathy: spectrum of disease. Am J Med. 1989;86(5):539-546.
doi - Cello JP, Chan MF. Long-term follow-up of endoscopic retrograde cholangiopancreatography sphincterotomy for patients with acquired immune deficiency syndrome papillary stenosis. Am J Med. 1995;99(6):600-603.
doi - Bouche H, Housset C, Dumont JL, Carnot F, Menu Y, Aveline B, Belghiti J, et al. AIDS-related cholangitis: diagnostic features and course in 15 patients. J Hepatol. 1993;17(1):34-39.
doi - Wilcox CM, Monkemuller KE. Hepatobiliary diseases in patients with AIDS: focus on AIDS cholangiopathy and gallbladder disease. Dig Dis. 1998;16(4):205-213.
doi pubmed - Yusuf TE, Baron TH. AIDS Cholangiopathy. Curr Treat Options Gastroenterol. 2004;7(2):111-117.
doi pubmed - Blumberg RS, Kelsey P, Perrone T, Dickersin R, Laquaglia M, Ferruci J. Cytomegalovirus- and Cryptosporidium-associated acalculous gangrenous cholecystitis. Am J Med. 1984;76(6):1118-1123.
doi - Margulis SJ, Honig CL, Soave R, Govoni AF, Mouradian JA, Jacobson IM. Biliary tract obstruction in the acquired immunodeficiency syndrome. Ann Intern Med. 1986;105(2):207-210.
doi pubmed - De Angelis C, Mangone M, Bianchi M, Saracco G, Repici A, Rizzetto M, Pellicano R. An update on AIDS-related cholangiopathy. Minerva Gastroenterol Dietol. 2009;55(1):79-82.
- Benhamou Y, Caumes E, Gerosa Y, Cadranel JF, Dohin E, Katlama C, Amouyal P, et al. AIDS-related cholangiopathy. Critical analysis of a prospective series of 26 patients. Dig Dis Sci. 1993;38(6):1113-1118.
doi pubmed - Devarbhavi H, Sebastian T, Seetharamu SM, Karanth D. HIV/AIDS cholangiopathy: clinical spectrum, cholangiographic features and outcome in 30 patients. J Gastroenterol Hepatol. 2010;25(10):1656-1660.
doi pubmed - Chen XM, Keithly JS, Paya CV, LaRusso NF. Cryptosporidiosis. N Engl J Med. 2002;346(22):1723-1731.
doi pubmed - Vakil NB, Schwartz SM, Buggy BP, Brummitt CF, Kherellah M, Letzer DM, Gilson IH, et al. Biliary cryptosporidiosis in HIV-infected people after the waterborne outbreak of cryptosporidiosis in Milwaukee. N Engl J Med. 1996;334(1):19-23.
doi pubmed - Naseer M, Dailey FE, Juboori AA, Samiullah S, Tahan V. Epidemiology, determinants, and management of AIDS cholangiopathy: A review. World J Gastroenterol. 2018;24(7):767-774.
doi pubmed - O'Hara SP, Small AJ, Gajdos GB, Badley AD, Chen XM, Larusso NF. HIV-1 Tat protein suppresses cholangiocyte toll-like receptor 4 expression and defense against Cryptosporidium parvum. J Infect Dis. 2009;199(8):1195-1204.
doi pubmed - Farman J, Brunetti J, Baer JW, Freiman H, Comer GM, Scholz FJ, Koehler RE, et al. AIDS-related cholangiopancreatographic changes. Abdom Imaging. 1994;19(5):417-422.
doi pubmed - Pol S, Romana C, Richard S, Carnot F, Dumont JL, Bouche H, Pialoux G, et al. Enterocytozoon bieneusi infection in acquired immunodeficiency syndrome-related sclerosing cholangitis. Gastroenterology. 1992;102(5):1778-1781.
doi - Hashmey R, Smith NH, Cron S, Graviss EA, Chappell CL, White AC, Jr. Cryptosporidiosis in Houston, Texas. A report of 95 cases. Medicine (Baltimore). 1997;76(2):118-139.
doi pubmed - Daly CA, Padley SP. Sonographic prediction of a normal or abnormal ERCP in suspected AIDS related sclerosing cholangitis. Clin Radiol. 1996;51(9):618-621.
doi - Da Silva F, Boudghene F, Lecomte I, Delage Y, Grange JD, Bigot JM. Sonography in AIDS-related cholangitis: prevalence and cause of an echogenic nodule in the distal end of the common bile duct. AJR Am J Roentgenol. 1993;160(6):1205-1207.
doi pubmed - Bilgin M, Balci NC, Erdogan A, Momtahen AJ, Alkaade S, Rau WS. Hepatobiliary and pancreatic MRI and MRCP findings in patients with HIV infection. AJR Am J Roentgenol. 2008;191(1):228-232.
doi pubmed - Tonolini M, Bianco R. HIV-related/AIDS cholangiopathy: pictorial review with emphasis on MRCP findings and differential diagnosis. Clin Imaging. 2013;37(2):219-226.
doi pubmed - Carucci LR, Halvorsen RA. Abdominal and pelvic CT in the HIV-positive population. Abdom Imaging. 2004;29(6):631-642.
doi pubmed - Forbes A, Blanshard C, Gazzard B. Natural history of AIDS related sclerosing cholangitis: a study of 20 cases. Gut. 1993;34(1):116-121.
doi pubmed - Chapman RW. Aetiology and natural history of primary sclerosing cholangitis—a decade of progress? Gut. 1991;32(12):1433-1435.
doi pubmed - Keaveny AP, Karasik MS. Hepatobiliary and pancreatic infections in AIDS: Part one. AIDS Patient Care STDS. 1998;12(5):347-357.
doi pubmed - Checkley W, White AC, Jr., Jaganath D, Arrowood MJ, Chalmers RM, Chen XM, Fayer R, et al. A review of the global burden, novel diagnostics, therapeutics, and vaccine targets for cryptosporidium. Lancet Infect Dis. 2015;15(1):85-94.
doi - Rossignol JF, Kabil SM, el-Gohary Y, Younis AM. Effect of nitazoxanide in diarrhea and enteritis caused by Cryptosporidium species. Clin Gastroenterol Hepatol. 2006;4(3):320-324.
doi pubmed - Amadi B, Mwiya M, Sianongo S, Payne L, Watuka A, Katubulushi M, Kelly P. High dose prolonged treatment with nitazoxanide is not effective for cryptosporidiosis in HIV positive Zambian children: a randomised controlled trial. BMC Infect Dis. 2009;9:195.
doi pubmed - Hewitt RG, Yiannoutsos CT, Higgs ES, Carey JT, Geiseler PJ, Soave R, Rosenberg R, et al. Paromomycin: no more effective than placebo for treatment of cryptosporidiosis in patients with advanced human immunodeficiency virus infection. AIDS Clinical Trial Group. Clin Infect Dis. 2000;31(4):1084-1092.
doi pubmed - Cabada MM, White AC, Jr. Treatment of cryptosporidiosis: do we know what we think we know? Curr Opin Infect Dis. 2010;23(5):494-499.
doi pubmed - Oku T, Maeda M, Waga E, Wada Y, Nagamachi Y, Fujita M, Suzuki Y, et al. Cytomegalovirus cholangitis and pancreatitis in an immunocompetent patient. J Gastroenterol. 2005;40(10):987-992.
doi pubmed - Molina JM, Oksenhendler E, Beauvais B, Sarfati C, Jaccard A, Derouin F, Modai J. Disseminated microsporidiosis due to Septata intestinalis in patients with AIDS: clinical features and response to albendazole therapy. J Infect Dis. 1995;171(1):245-249.
doi pubmed - Deodhar L, Maniar JK, Saple DG. Cyclospora infection in acquired immunodeficiency syndrome. J Assoc Physicians India. 2000;48(4):404-406.
- Sifuentes-Osornio J, Porras-Cortes G, Bendall RP, Morales-Villarreal F, Reyes-Teran G, Ruiz-Palacios GM. Cyclospora cayetanensis infection in patients with and without AIDS: biliary disease as another clinical manifestation. Clin Infect Dis. 1995;21(5):1092-1097.
doi pubmed - Lagrange-Xelot M, Porcher R, Sarfati C, de Castro N, Carel O, Magnier JD, Delcey V, et al. Isosporiasis in patients with HIV infection in the highly active antiretroviral therapy era in France. HIV Med. 2008;9(2):126-130.
doi pubmed - Castiella A, Iribarren JA, Lopez P, Arrizabalaga J, Rodriguez F, von Wichmann MA, Arenas JI. Ursodeoxycholic acid in the treatment of AIDS-associated cholangiopathy. Am J Med. 1997;103(2):170-171.
- Casseb J, Fonseca LA, Veiga AP, de Almeida A, Bueno A, Ferez AC, Gonsalez CR, et al. AIDS incidence and mortality in a hospital-based cohort of HIV-1-seropositive patients receiving highly active antiretroviral therapy in Sao Paulo, Brazil. AIDS Patient Care STDS. 2003;17(9):447-452.
doi pubmed - Palella FJ, Jr., Delaney KM, Moorman AC, Loveless MO, Fuhrer J, Satten GA, Aschman DJ, et al. Declining morbidity and mortality among patients with advanced human immunodeficiency virus infection. HIV Outpatient Study Investigators. N Engl J Med. 1998;338(13):853-860.
doi pubmed - Maggi P, Larocca AM, Quarto M, Serio G, Brandonisio O, Angarano G, Pastore G. Effect of antiretroviral therapy on cryptosporidiosis and microsporidiosis in patients infected with human immunodeficiency virus type 1. Eur J Clin Microbiol Infect Dis. 2000;19(3):213-217.
doi pubmed - Miao YM, Awad-El-Kariem FM, Franzen C, Ellis DS, Muller A, Counihan HM, Hayes PJ, et al. Eradication of cryptosporidia and microsporidia following successful antiretroviral therapy. J Acquir Immune Defic Syndr. 2000;25(2):124-129.
doi pubmed - Carr A, Marriott D, Field A, Vasak E, Cooper DA. Treatment of HIV-1-associated microsporidiosis and cryptosporidiosis with combination antiretroviral therapy. Lancet. 1998;351(9098):256-261.
doi - Datta J, Shafi BM, Drebin JA. Extrahepatic cholangiocarcinoma developing in the setting of AIDS cholangiopathy. Am Surg. 2013;79(3):321-322.
doi pubmed - Charlier C, Lecuit M, Furco A, Estavoyer JM, Lafeuillade A, Dupont B, Lortholary O, et al. Cholangiocarcinoma in HIV-Infected patients with a history of Cholangitis. J Acquir Immune Defic Syndr. 2005;39(2):253-255.
- Mangeya N, Mafukidze AT, Pascoe M, Mbuwayesango B, Madziva D, Ndlovu N, Corbett EL, et al. Cholangiocarcinoma presenting in an adolescent with vertically acquired HIV infection. Int J STD AIDS. 2008;19(10):717-718.
doi pubmed - Ko WF, Cello JP, Rogers SJ, Lecours A. Prognostic factors for the survival of patients with AIDS cholangiopathy. Am J Gastroenterol. 2003;98(10):2176-2181.
doi pubmed
This article is distributed under the terms of the Creative Commons Attribution Non-Commercial 4.0 International License, which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Journal of Medical Cases is published by Elmer Press Inc.


