| Journal of Medical Cases, ISSN 1923-4155 print, 1923-4163 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Med Cases and Elmer Press Inc |
| Journal website https://www.journalmc.org |
Case Report
Volume 14, Number 8, August 2023, pages 271-276
Tuberculous Pericarditis Presenting as Cardiac Tamponade: Role of Echocardiography
Nazima Khatuna, Yonatan Akivisa, c , Beisi Jia, Harshith P. Chandrakumara, Inna Bukharovichb, Sabu Johnb
aDivision of Cardiovascular Medicine, Department of Internal Medicine, College of Medicine, SUNY Downstate-Health Science University, Brooklyn, NY 11203, USA
bKings County Hospital Center, Brooklyn, NY 11203, USA
cCorresponding Author: Yonatan Akivis, Division of Cardiovascular Medicine, Department of Medicine, College of Medicine, SUNY Downstate-Health Science University, Brooklyn, NY 11203, USA
Manuscript submitted May 24, 2023, accepted July 6, 2023, published online August 28, 2023
Short title: TBP Presenting as Cardiac Tamponade
doi: https://doi.org/10.14740/jmc4119
| Abstract | ▴Top |
Tuberculous pericarditis, a rare but potentially lethal manifestation of tuberculosis, poses diagnostic and therapeutic challenges in clinical practice. Its nonspecific clinical presentation often mimics other conditions, leading to delayed or missed diagnoses. We report a 25-year-old male with no past medical history, who presented with nonspecific symptoms such as fatigue, weight loss, body aches, and dyspnea. An electrocardiogram showed low voltage QRS complex with electrical alternans, and transthoracic echocardiography (TTE) showed large pericardial effusion with tamponade physiology with right ventricular diastolic collapse, the collapse of the right atrium and the inferior vena cava was dilated with a respiratory variation of less than 50%. The diagnosis of tuberculous pericarditis was made based on clinical presentation, imaging, and laboratory findings, including a positive QuantiFERON-TB gold test and pericardial fluid analysis, despite negative cultures. This case highlights the significance of considering tuberculosis in the differential diagnosis of pericardial effusion and underscores the role of imaging and laboratory investigations in diagnosis. Management of tuberculous pericarditis involves a combination of antituberculous chemotherapy, pericardiocentesis, and corticosteroids. Despite its rarity, tuberculous pericarditis carries a high mortality rate and can present as cardiac tamponade, as illustrated in our case. This underscores the need for high clinical suspicion, especially in high-risk populations, for timely diagnosis and initiation of treatment.
Keywords: Pericardial effusion; Cardiac tamponade; Tuberculous pericarditis; Transthoracic echocardiography
| Introduction | ▴Top |
Tuberculosis, an infectious disease predominantly caused by Mycobacterium tuberculosis, is a major global health concern. Despite efforts towards eradication, it continues to burden many regions of the world, particularly in low- and middle-income countries [1]. Primarily a pulmonary disease, tuberculosis can disseminate to involve other parts of the body, demonstrating a wide array of clinical manifestations [2].
One such extrapulmonary manifestation is tuberculous pericarditis (TBP), a form of tuberculosis that affects the pericardium, the dual-layered sac that envelops the heart. Although tuberculosis is a prevalent disease worldwide, pericardial involvement is relatively uncommon, accounting for approximately 1-2% of all cases of tuberculosis [3, 4]. The pathogenesis typically involves the hematogenous spread or direct extension of tubercle bacilli from a neighboring focus of infection to the pericardium. Symptoms are usually insidious in onset, with patients often presenting with chest discomfort, dyspnea, and fatigue. The vague and nonspecific nature of these symptoms can pose diagnostic challenges.
In certain cases, TBP can progress to cardiac tamponade, a severe, life-threatening condition resulting from the rapid accumulation of pericardial fluid. This impedes cardiac filling, leading to hemodynamic compromise. Although classical presentation includes hypotension, distended neck veins, and muffled heart sounds, known as Beck’s triad, atypical presentations are not uncommon, further complicating the diagnostic process [5].
Echocardiography plays a crucial role in this context. This noninvasive imaging modality provides key insights into cardiac structure and function, aiding in detecting pericardial effusions, assessing their hemodynamic impact, and guiding pericardiocentesis if required [5, 6].
This case report highlights an unusual presentation of TBP leading to cardiac tamponade, illustrating the vital role of echocardiography in its diagnosis and management. Our aim is to enhance understanding of this rare manifestation of widespread disease and emphasize the importance of early recognition and timely intervention.
| Case Report | ▴Top |
Investigations
The patient is a 25-year-old male with no significant past medical history. He presented to the emergency department (ED) with a 4-month history of progressively worsening body aches, fatigue, and weight loss. Four days prior to presentation, the patient started experiencing dyspnea on exertion, accompanied by bilateral ankle swelling. Upon review of systems, he endorsed a history of intermittent fevers, chills, generalized malaise, unexplained weight loss, nausea, non-bloody non-bilious vomiting, and abdominal distention.
Upon initial evaluation in the ED, his vital signs were as follows: afebrile with a temperature of 36.9 °C, blood pressure of 114/90 mm Hg, pulse rate of 97 beats per minute, respiratory rate of 18 breaths per minute, and oxygen saturation of 97% on room air. The physical examination revealed an alert and oriented patient with decreased breath sounds in the right lower lung fields. Cardiac auscultation indicated tachycardia with distant heart sounds. His abdomen was distended with diffuse tenderness. A musculoskeletal examination noted pitting edema up to the level of both ankles. The remainder of his examination was unremarkable. The patient’s social history was negative for smoking, alcoholism, or drug abuse. Furthermore, there were no recent sick contacts. The patient traveled home to Haiti 8 months prior to the presentation.
Diagnosis
Lab values on the initial presentation are displayed in Table 1.
 Click to view | Table 1. Laboratory Values |
Electrocardiogram (EKG) showed normal sinus rhythm with low voltage QRS complexes and electrical alternans (Fig. 1).
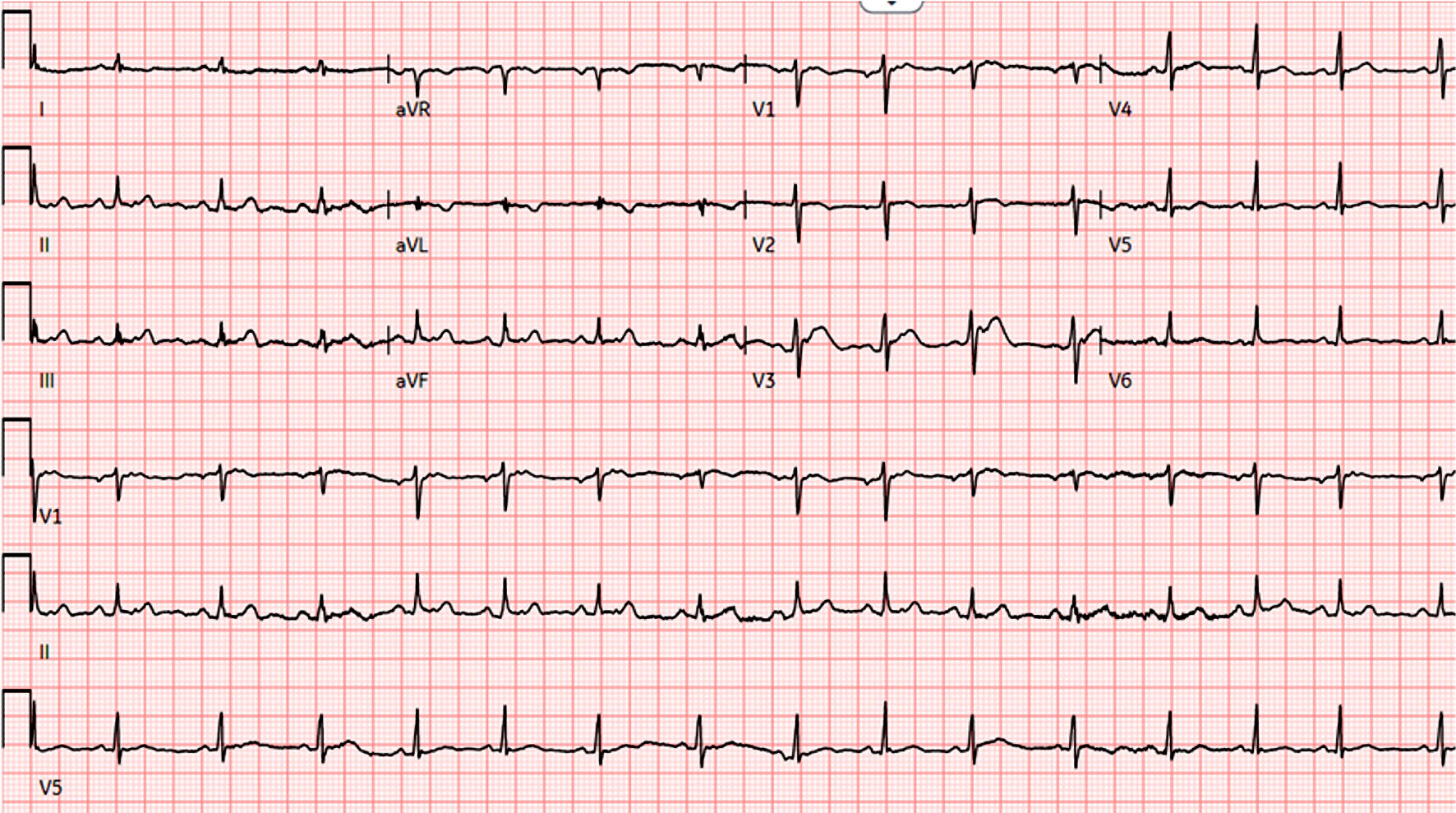 Click for large image | Figure 1. EKG shows low voltage QRS complexes and electrical alternans. EKG: electrocardiogram. |
A chest X-ray showed a moderate right pleural effusion and cardiomegaly. A computed tomography (CT) scan of the chest, abdomen, and pelvis revealed a large pericardial effusion, thickened pulmonary fissures, and intraabdominal ascites (Fig. 2).
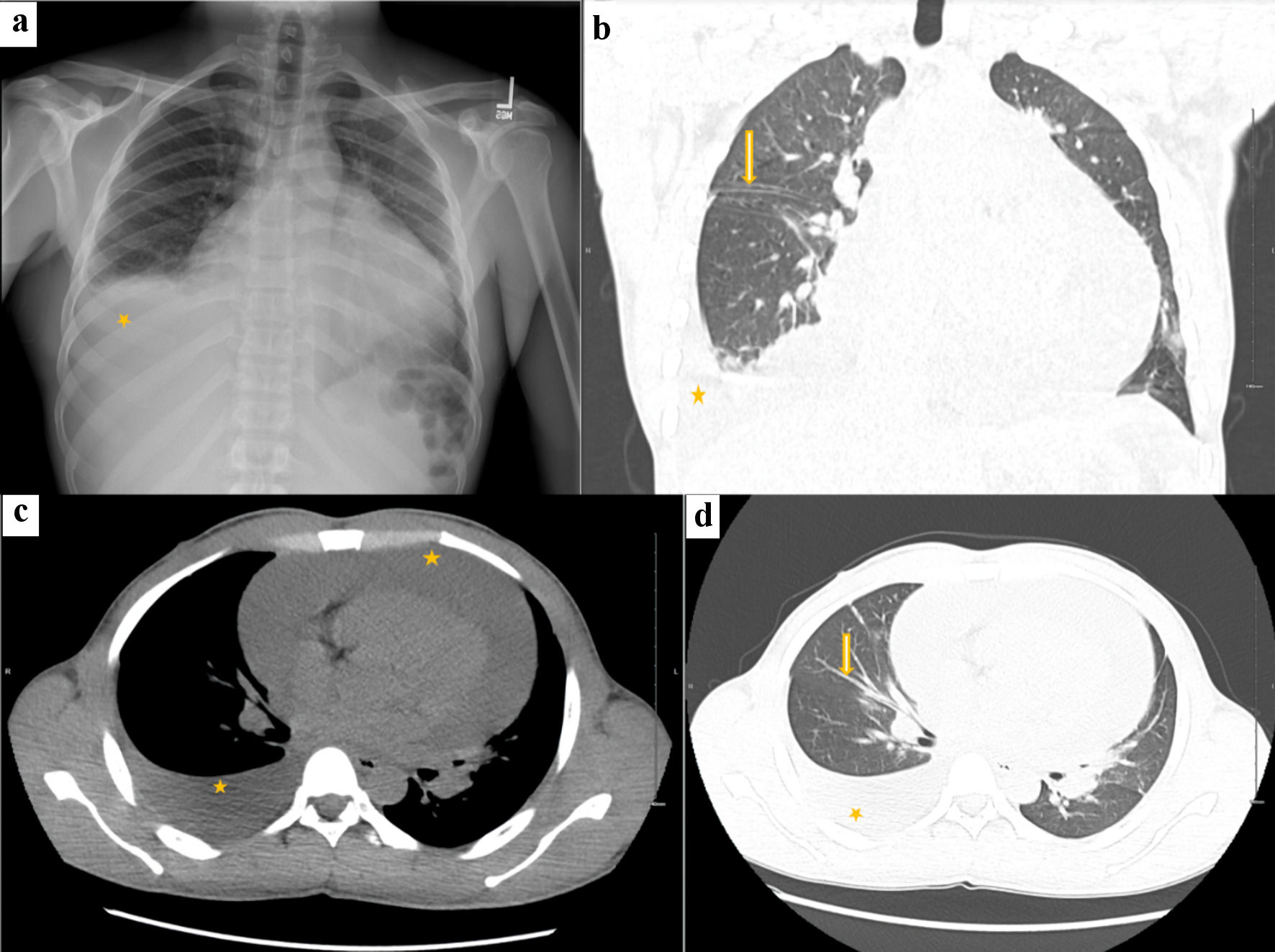 Click for large image | Figure 2. Chest X-ray and CT chest. Chest X-ray (a) shows cardiomegaly with right-sided pleural effusion (asterisk). CT chest (b) coronal section shows a globular heart, volume overload with thickened pulmonary fissures (arrow) and interlobular septa, and moderate right-sided pleural effusion (asterisk). CT chest axial section (c) mediastinal window shows large pericardial effusion (asterisk) with moderate right-sided pleural effusion (asterisk), and (d) lung window shows volume overload with increased bronchovascular markings (arrow) and moderate right-sided pleural effusion (asterisk). CT: computed tomography. |
Transthoracic echocardiography confirmed a large pericardial effusion with right ventricular (RV) diastolic collapse, consistent with tamponade physiology (Fig. 3).
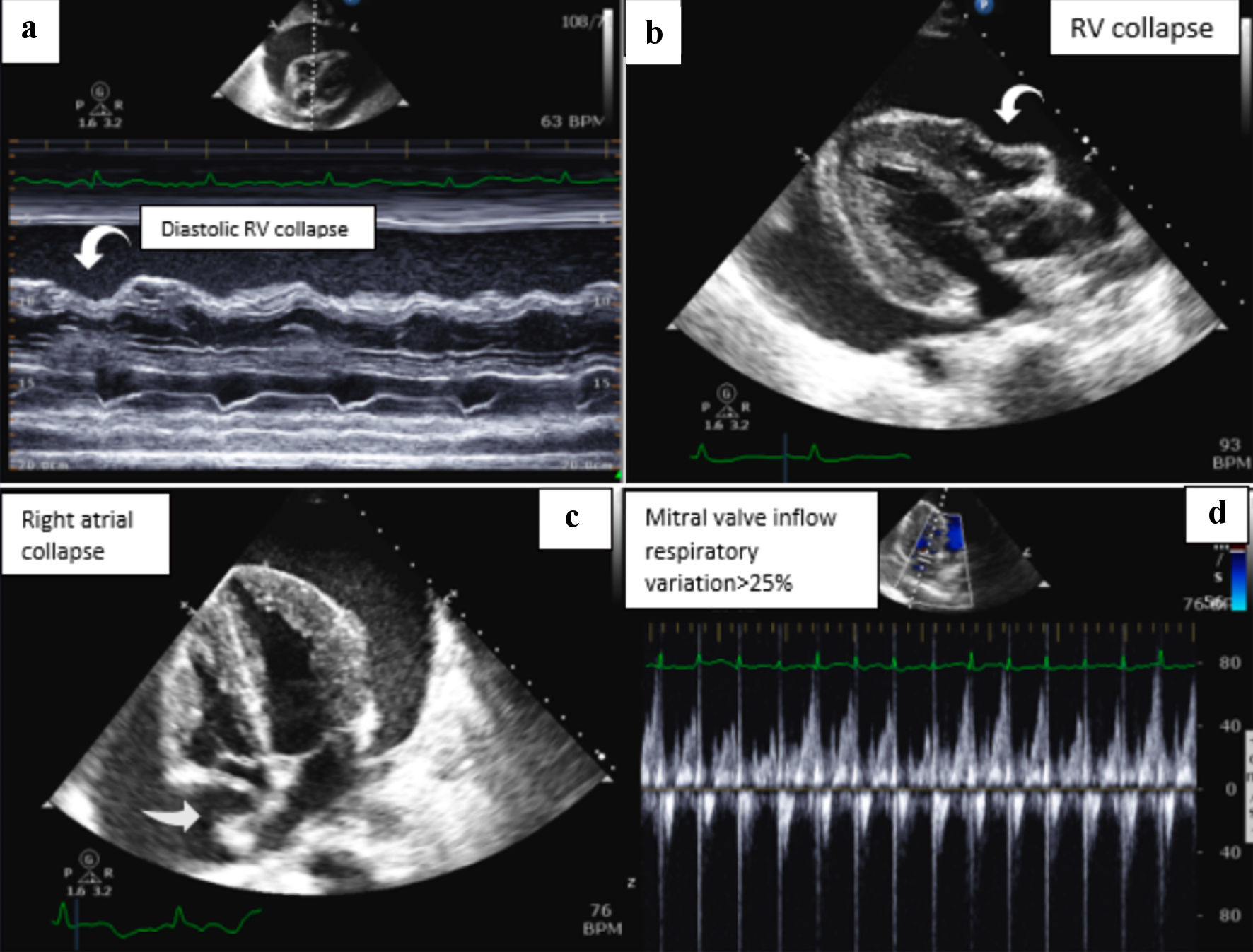 Click for large image | Figure 3. TTE images show the following findings: (a) M-mode with large pericardial effusion and right ventricular (RV) diastolic collapse. (b) Parasternal long axis view consistent with RV diastolic collapse. (c) The collapse of the right atrium. (d) Mitral valve inflow velocity with respiratory variation greater than 25%. TTE: transthoracic echocardiography. |
The patient underwent pericardiocentesis, during which 1,500 mL of bloody fluid was drained. The pericardial fluid analysis was predominantly lymphocytic, but cultures, including acid-fast bacilli (AFB), were negative. The adenosine deaminase (ADA) level was 43 U/L (reference range 0 - 30). An extensive rheumatological workup, including antinuclear antibody, rheumatoid factor, anti-neutrophilic cytoplasmic autoantibody, and beta-2 glycoprotein, was unremarkable. However, the QuantiFERON-TB gold test returned positive.
Treatment
Given no other source of infection or autoimmune etiology, the patient was diagnosed with tuberculosis and the pericardial effusion was attributed to his infection. The patient was started on daily doses of rifampin 600 mg, isoniazid 300 mg, pyrazinamide 1.5 g, ethambutol 1.2 g, and vitamin B6 50 mg (RIPE). He was discharged on directly observed therapy (DOT) and a prednisone taper (60 mg for 4 weeks, 30 mg for 4 weeks, 15 mg for 2 weeks, and 5 mg for 1 week) given his high risk of inflammatory complications.
Follow-up and outcomes
The patient has since remained stable, feeling well, with no signs of new infectious disease or pericardial effusion return.
| Discussion | ▴Top |
TBP, though rare in developed countries with an occurrence rate of roughly 1%, presents significant clinical challenges due to its high mortality rate [1, 7]. The disease’s protean presentations, which include pericardial effusion, constrictive pericarditis, and a complex scenario combining both effusion and constriction, add to the diagnostic conundrum [7, 8].
TBP characteristically develops insidiously, often clouded by a myriad of nonspecific symptoms such as fever, night sweats, fatigue, and weight loss, thereby complicating early detection and treatment [9-11]. Patients often complain of chest pain, cough, and dyspnea, although severe acute-onset pericardial pain is rare.
Understanding the pathophysiology of TBP can provide significant insights into its insidious onset and the subsequent presentation of symptoms. The Mycobacterium tuberculosis bacillus, responsible for tuberculosis, has a predilection for oxygen-rich tissues, which explains why the lungs are a primary site of infection [3, 6]. However, it can also reach the pericardium through hematogenous spread, lymphatic dissemination, or direct extension from a contiguous focus of infection, such as the lungs, mediastinal lymph nodes, or the spine. When the bacilli reach the pericardium, they incite an inflammatory response leading to the formation of granulomas - nodular inflammatory lesions that characterize tuberculosis. As these granulomas grow, they erode the pericardium, causing an influx of protein-rich fluid into the pericardial space, resulting in pericardial effusion [3, 6]. In some cases, the bacilli further induce a caseating necrosis within the granulomas, leading to the release of more inflammatory and necrotic debris into the pericardial space. This can result in a thicker, more “constrictive” pericardial effusion and may ultimately progress to constrictive pericarditis [3, 6, 8-10]. It is noteworthy that the progression from pericardial effusion to constrictive pericarditis is not inevitable and largely depends on the host’s immune response. A strong and rapid immune response can contain the infection and prevent the development of pericardial effusion or progression to constrictive pericarditis. However, in individuals with a compromised immune system, such as those with human immunodeficiency virus (HIV)/acquired immunodeficiency syndrome (AIDS), the infection may progress more aggressively.
Echocardiography plays a pivotal role in the diagnosis and management of TBP. This noninvasive imaging modality enables clinicians to visually assess the size and extent of the pericardial effusion, detect any thickening of the visceral pericardium, and identify signs of cardiac tamponade. Moreover, echocardiography can monitor the response to therapy by tracking changes in effusion size and other pericardial features. Advanced echocardiographic techniques can provide even deeper insights. For instance, two-dimensional echocardiography and M-mode color Doppler can help identify features of cardiac tamponade, including diastolic collapse of the anterior RV free wall and right atrium, dilatation of the inferior vena cava, and large respiratory fluctuations in mitral and tricuspid inflow velocities. These findings are crucial in determining the need for urgent interventions like pericardiocentesis. Additionally, echocardiography can offer longitudinal monitoring of patients with TBP, facilitating the timely identification of potential complications such as constrictive pericarditis. In this case, echocardiographic findings, including pericardial thickening, pericardial calcification, and signs of impaired ventricular filling, can inform the need for potential surgical intervention [12-14].
Given the well-documented association between TBP and HIV, it is paramount that HIV testing, sputum smear, and culture be considered in the diagnostic pathway of all suspected TBP patients. Moreover, the onset of large pericardial effusion or tamponade, or when there is a strong clinical suspicion of tuberculosis, the necessity of pericardiocentesis becomes apparent. This invasive procedure enables the collection of pericardial fluid for further analysis, which typically reveals an exudative effusion characterized by a high protein and increased leukocyte count, primarily constituted by lymphocytes and monocytes [3, 15, 16]. Clinicians should also be aware that elevated levels of ADA (> 40 U/L) and interferon (IFN)-gamma (> 200 pg/L) in the pericardial effusion are strong indicators of TBP.
The gold standard for confirming TBP diagnosis remains the identification of tubercle bacilli in the pericardial fluid or tissue or the presence of caseating granulomata on histological examination of the pericardium [7]. However, possible TBP diagnosis can be inferred from several other clinical features such as the evidence of pericarditis in a patient with tuberculosis demonstrated elsewhere in the body, lymphocytic pericardial exudate with elevated ADA activity, or a noticeable response to antituberculosis chemotherapy [15-17]. The comprehensive treatment of TBP is three-pronged: 1) Attacking the underlying tuberculosis infection, usually through a regimen of rifampicin, isoniazid, ethambutol, and pyrazinamide for a minimum of 2 months, succeeded by isoniazid and rifampin for an aggregate of 6-month duration; 2) Performing pericardiocentesis to manage pericardial effusion and potential tamponade; and 3) Administering corticosteroids to preemptively combat constrictive complications that can arise [3].
Each part of this therapeutic strategy plays a vital role in combating TBP. Addressing the tuberculosis infection aggressively with a potent antibiotic regimen forms the cornerstone of TBP treatment. Rifampicin, isoniazid, ethambutol, and pyrazinamide, which comprise the first-line anti-tuberculosis medications, are typically administered concurrently for an intensive 2-month period. The treatment then follows for 4 more months with isoniazid and rifampin [1]. In situations with large pericardial effusions or tamponade, pericardiocentesis can be lifesaving. It also provides the opportunity for further analysis of the pericardial fluid, which could lead to a definitive diagnosis in some cases.
The role of corticosteroids in the treatment of TBP warrants further elaboration. These potent anti-inflammatory drugs are used with the aim of preventing the progression to constrictive pericarditis, a severe complication that can lead to diminished heart function and even death. Corticosteroids function by reducing the inflammatory response in the pericardial space, thereby potentially preventing fibrosis and consequent constriction [3]. However, their use must be balanced with the potential for side effects, and thus they are typically employed when there is a high risk of constriction or when other treatment methods have not produced satisfactory results.
Conclusions
TBP, despite its rarity in developed nations, signifies a pressing concern due to its insidious nature, high mortality rate, and intricacy in diagnosis and treatment. Its vague symptomatology and gradual onset necessitate keen clinical vigilance, particularly in regions with high tuberculosis prevalence or among populations vulnerable to tuberculosis infection.
Advancements in diagnostic tools, including echocardiography and CT scanning, as well as pericardial fluid analysis, have considerably enhanced our ability to detect this disease. Nevertheless, conclusive diagnosis often hinges upon the identification of tubercle bacilli or histological evidence of caseating granulomata in the pericardium.
Effective management of TBP involves a multifaceted approach, including antituberculous chemotherapy, therapeutic pericardiocentesis, and judicious use of corticosteroids to prevent constrictive sequelae. A proactive approach, timely diagnosis, and appropriate interventions are essential in improving patient prognosis and preventing potential complications.
Learning points
TBP should be considered as a potential diagnosis in patients presenting with insidious onset of symptoms such as weight loss, night sweats, fatigue, and dyspnea, especially in individuals from regions where tuberculosis is prevalent or in high-risk populations.
Diagnostic modalities like echocardiography and CT scan can provide valuable information in suspecting TBP. In particular, echocardiographic evidence of pericardial effusion, thickening of the visceral pericardium, or diastolic collapse of anterior RV free wall and right atrium can be suggestive of this condition.
Laboratory investigations including sputum smear and culture, HIV testing, and pericardial fluid studies play a crucial role in establishing a diagnosis. Specifically, a lymphocytic exudative pericardial effusion with high ADA activity or IFN-gamma levels can hint towards TBP.
Acknowledgments
None to declare.
Financial Disclosure
None to disclose.
Conflict of Interest
None to disclose.
Informed Consent
Written consent was obtained.
Author Contributions
All authors contributed to the writing/editing part of this paper. Nazima Khatun, Yonatan Akivis, Beisi Ji, and Harshith P Chandrakumar wrote the manuscript. Sabu John and Inna Bukharovich edited and guided us in writing this manuscript.
Data Availability
The authors declare that data supporting the findings of this study are available within the article.
Abbreviations
TBP: tuberculous pericarditis; HIV: human immunodeficiency virus; EKG: electrocardiogram; LV: left ventricle; RV: right ventricle; IFN: interferon; ADA: adenosine deaminase; TTE: transthoracic echocardiography
| References | ▴Top |
- Garcia-Garcia-de-Paredes A, Rodriguez-de-Santiago E, Aguilera-Castro L, Ferre-Aracil C, Lopez-Sanroman A. [Fecal microbiota transplantation]. Gastroenterol Hepatol. 2015;38(3):123-134.
doi pubmed - Zumla A, Raviglione M, Hafner R, von Reyn CF. Tuberculosis. N Engl J Med. 2013;368(8):745-755.
doi pubmed - Mayosi BM, Burgess LJ, Doubell AF. Tuberculous pericarditis. Circulation. 2005;112(23):3608-3616.
doi pubmed - Syed FF, Mayosi BM. A modern approach to tuberculous pericarditis. Prog Cardiovasc Dis. 2007;50(3):218-236.
doi pubmed - Roy CL, Minor MA, Brookhart MA, Choudhry NK. Does this patient with a pericardial effusion have cardiac tamponade? JAMA. 2007;297(16):1810-1818.
doi pubmed - Lange RA, Hillis LD. Clinical practice. Acute pericarditis. N Engl J Med. 2004;351(21):2195-2202.
doi pubmed - Adler Y, Charron P, Imazio M, Badano L, Baron-Esquivias G, Bogaert J, Brucato A, et al. 2015 ESC Guidelines for the diagnosis and management of pericardial diseases: The Task Force for the Diagnosis and Management of Pericardial Diseases of the European Society of Cardiology (ESC)Endorsed by: The European Association for Cardio-Thoracic Surgery (EACTS). Eur Heart J. 2015;36(42):2921-2964.
doi pubmed pmc - Filardo TD, Feng PJ, Pratt RH, Price SF, Self JL. Tuberculosis - United States, 2021. MMWR Morb Mortal Wkly Rep. 2022;71(12):441-446.
doi pubmed pmc - Mathiasen VD, Frederiksen CA, Wejse C, Poulsen SH. A clinical case of tuberculosis with transient constrictive pericarditis and perimyocarditis. Echo Res Pract. 2019;6(3):K7-K12.
doi pubmed pmc - Amado Garzon SB, et al. Tuberculosis extrapulmonar: un reto clinico vigente. Universitas Medica. 2020;61(4):66-79.
- Reuter H, Burgess L, van Vuuren W, Doubell A. Diagnosing tuberculous pericarditis. QJM. 2006;99(12):827-839.
doi pubmed - Reuter H, Burgess LJ, Doubell AF. Role of chest radiography in diagnosing patients with tuberculous pericarditis. Cardiovasc J S Afr. 2005;16(2):108-111.
pubmed - Levine MJ, Lorell BH, Diver DJ, Come PC. Implications of echocardiographically assisted diagnosis of pericardial tamponade in contemporary medical patients: detection before hemodynamic embarrassment. J Am Coll Cardiol. 1991;17(1):59-65.
doi pubmed - Reydel B, Spodick DH. Frequency and significance of chamber collapses during cardiac tamponade. Am Heart J. 1990;119(5):1160-1163.
doi pubmed - Feigenbaum H, Zaky A, Grabhorn LL. Cardiac motion in patients with pericardial effusion. A study using reflected ultrasound. Circulation. 1966;34(4):611-619.
doi pubmed - Koh KK, Kim EJ, Cho CH, Choi MJ, Cho SK, Kim SS, Kim MH, et al. Adenosine deaminase and carcinoembryonic antigen in pericardial effusion diagnosis, especially in suspected tuberculous pericarditis. Circulation. 1994;89(6):2728-2735.
doi pubmed - Burgess LJ, Reuter H, Carstens ME, Taljaard JJ, Doubell AF. The use of adenosine deaminase and interferon-gamma as diagnostic tools for tuberculous pericarditis. Chest. 2002;122(3):900-905.
doi pubmed
This article is distributed under the terms of the Creative Commons Attribution Non-Commercial 4.0 International License, which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Journal of Medical Cases is published by Elmer Press Inc.


