| Journal of Medical Cases, ISSN 1923-4155 print, 1923-4163 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Med Cases and Elmer Press Inc |
| Journal website https://www.journalmc.org |
Case Report
Volume 14, Number 1, January 2023, pages 7-12
Uterine Inversion Secondary to Endometrial Carcinoma
Wanxuan Wanga, c, Junjie Wangb
aDepartment of Obstetrics and Gynaecology, KK Women’s and Children’s Hospital, Singapore 229899, Singapore
bDepartment of Gynaecological Oncology, KK Women’s and Children’s Hospital, Singapore 229899, Singapore
cCorresponding Author: Wanxuan Wang, Department of Obstetrics and Gynaecology, KK Women’s and Children’s Hospital, Singapore 229899, Singapore
Manuscript submitted November 28, 2022, accepted December 15, 2022, published online December 30, 2022
Short title: Uterine Inversion Caused by Endometrial Carcinoma
doi: https://doi.org/10.14740/jmc4023
| Abstract | ▴Top |
Uterine inversion may be puerperal or non-puerperal. Puerperal uterine inversion is a rare obstetrics complication, with an estimated incidence of 1 in 30,000 deliveries. The occurrence of non-puerperal uterine inversion is even rarer such that there is no good estimate of its incidence. It is challenging to make the diagnosis of non-puerperal uterine inversion and a high index of suspicion needs to be present. Malignancy is an uncommon cause for non-puerperal uterine inversion, but it is important to keep it in mind so as to counsel patients appropriately and prevent repeat surgery wherever possible. There are also unique complexities in the assessment of a virgo intacta patient which needs to be overcome through the use of different approaches in physical examination and imaging. In this case report, there is a unique interplay of multiple complicating factors in a virgo intacta patient presenting with abnormal uterine bleeding secondary to a malignant fibroid polyp that had prolapsed out of the vagina causing uterine inversion. She eventually required two open surgeries in the management of her condition.
Keywords: Non-puerperal uterine inversion; Prolapsed fibroid polyp; Endometrial carcinoma; Huntington’s method
| Introduction | ▴Top |
Uterine inversion, which is when the uterine fundus sinks into the endometrial cavity and turns the uterus partially or completely inside out, may be puerperal or non-puerperal. Puerperal uterine inversion is a rare obstetrics complication which usually occurs after the baby has been delivered. It has an estimated incidence of 1 in 30,000 deliveries [1]. The occurrence of non-puerperal uterine inversion is even rarer such that there is no good estimate of its incidence [2]. The most common causes for non-puerperal uterine inversion are polypoidal uterine tumors, of which up to 85% are benign leiomyomas [2-4]. The remaining causes are leiomyosarcoma, endometrial carcinoma, rhabdomyosarcoma, malignant mixed Mullerian tumors, endometrial stromal sarcoma, and endometrial polyps [5, 6]. Malignancy may account for up to 30% of cases [7].
It is challenging to make the diagnosis of non-puerperal uterine inversion as it rarely happens, and a high index of suspicion needs to be present in order to consider it as an important differential. On top of history and physical examination findings, ultrasound and magnetic resonance imaging (MRI) may aid in the diagnosis [6]. As malignancy may be the cause of non-puerperal uterine inversion [7], it is imperative to keep it in mind when counseling patients regarding management of suspected uterine inversion to prevent the need for a second surgery wherever possible. In this case report, we discuss a case of uterine inversion in a young patient that was eventually diagnosed to be secondary to endometrioid adenocarcinoma of the uterus. The case also presents the unique challenges in the assessment of a virgo intacta patient, with the state of “virgo intacta” referring to women who have never had sexual intercourse previously.
| Case Report | ▴Top |
Investigations
A 30-year-old single woman presented to our emergency Women’s Clinic with symptomatic anemia and a 4-year history of prolonged periods. She had regular 30-day cycles with periods lasting for 14 to 21 days. She considered her menstrual flow to be normal until a month ago when she noticed that the volume had increased significantly. She had been given oral norethisterone 10 mg, twice a day (BID) after 17 days of heavy bleeding, but this did not stop her period which continued beyond its usual 21-day duration. She also started experiencing giddiness, dyspnea on exertion, and palpitations.
On examination, her heart rate was 99 beats/min, blood pressure was 115/78 mm Hg, and her temperature was 37.6 °C. There was conjunctival pallor. Her abdomen was soft and non-tender. An internal examination was not performed as she was virgo intacta.
Diagnosis
Blood tests revealed a hemoglobin level of 5.2 g/dL. She was admitted from the emergency Women’s Clinic for blood transfusion and further investigations. A pelvic ultrasound scan performed with a transabdominal probe revealed that there was a poorly defined mass within the central part of the uterus measuring 8.3 × 3.9 × 4.2 cm. Blood vessels could be seen extending from the uterine fundus and the anterior wall of the uterus into the mass. The mass appeared to be extending into the cervix and probably upper vagina too. These findings were suggestive of a fibroid polyp but as the uterine fundus was not well seen, inversion of the uterine fundus could not be ruled out (Figs. 1, 2).
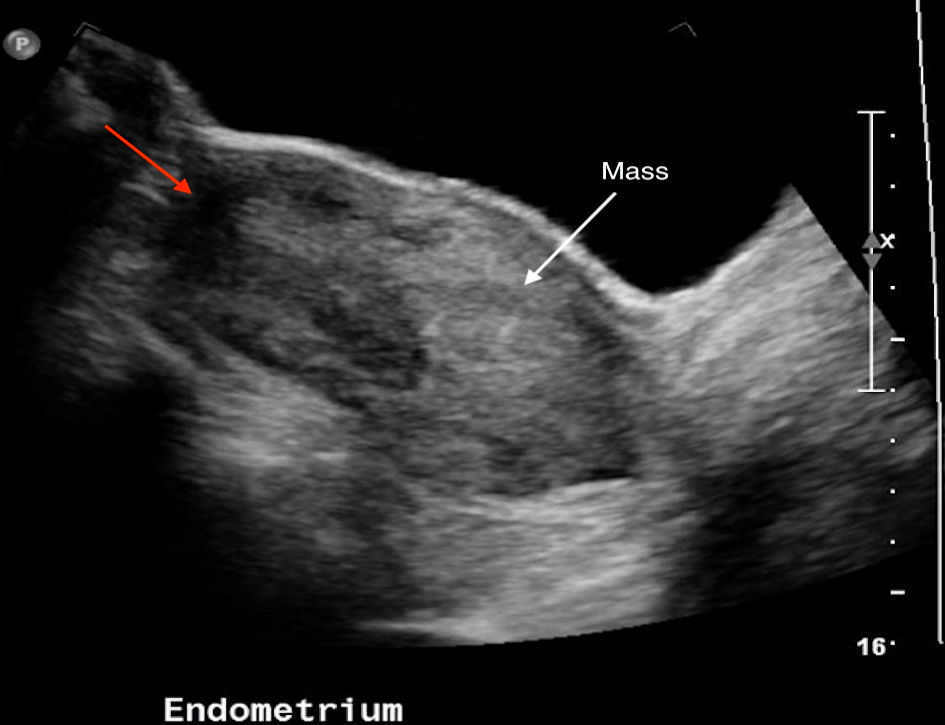 Click for large image | Figure 1. Ultrasound image with white arrow pointing to poorly defined mass within endometrial cavity. Red arrow indicates area where uterine fundus should be but was not well seen. |
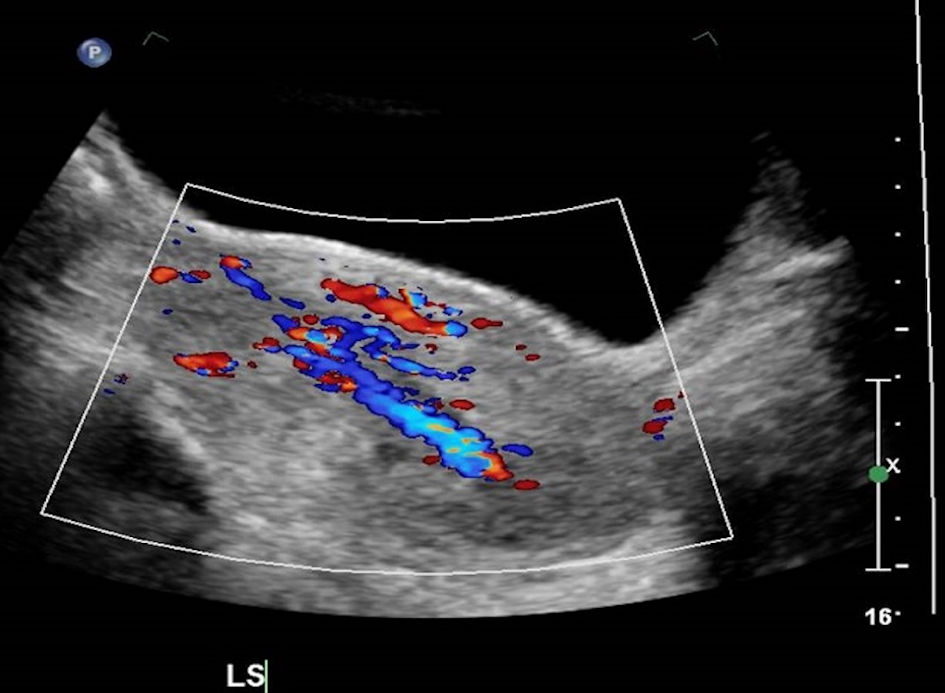 Click for large image | Figure 2. Ultrasound image with Doppler blood flow showing blood vessels extending from the uterine fundus and anterior wall of the uterus into the mass. |
Treatment
The patient was initially planned for hysteroscopy, dilatation, and curettage, with view for transcervical resection of polyp/myoma (TCRP/TCRM). Oral misoprostol was administered in preparation for the surgery.
However, while she was awaiting the surgery, there was an increase in per vaginal bleeding. On examination, there was a prolapsed mass measuring 5 cm at the introitus with a 3-cm thick stalk (Fig. 3). The prolapsed mass was actively bleeding then. She was resuscitated and counseled for emergency hysteroscopy, dilatation, and curettage, TCRM with the view for hysterectomy should there be persistent bleeding. She was placed on close monitoring until the time of her operation.
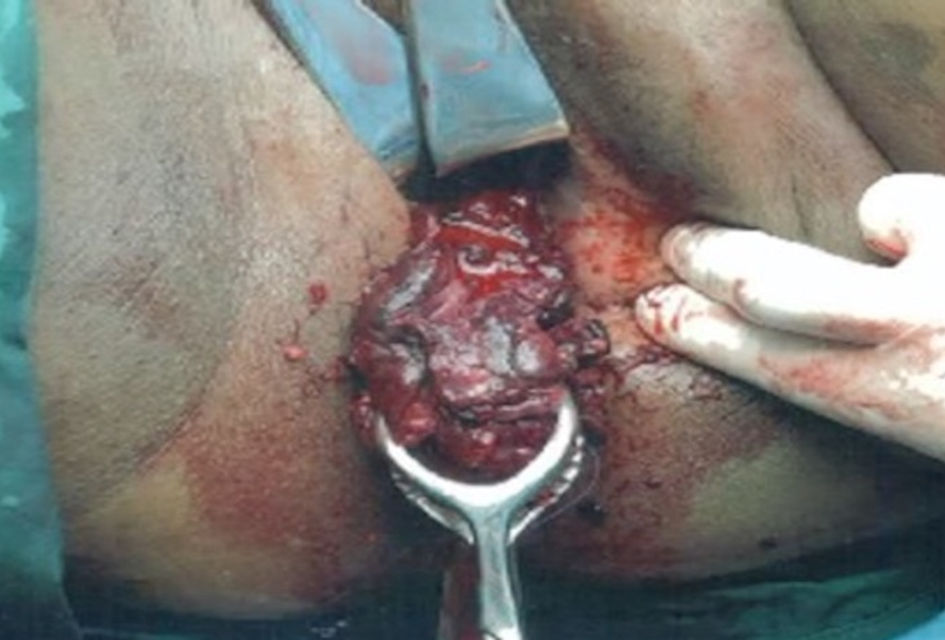 Click for large image | Figure 3. Prolapsed mass at introitus. |
Intraoperatively, hysteroscopy showed that there was a 4 × 6 cm mass with its pedicle attached to the uterine fundus that had prolapsed out of her uterus. The mass was actively bleeding, and it looked like a fibroid polyp. Under hysteroscopic guidance, the mass was ligated with a Vicryl suture before being resected with diathermy. It was then that uterine inversion was suspected.
Decision was made for diagnostic laparoscopy, which confirmed that there was complete uterine inversion (Fig. 4). Adnexa were normal.
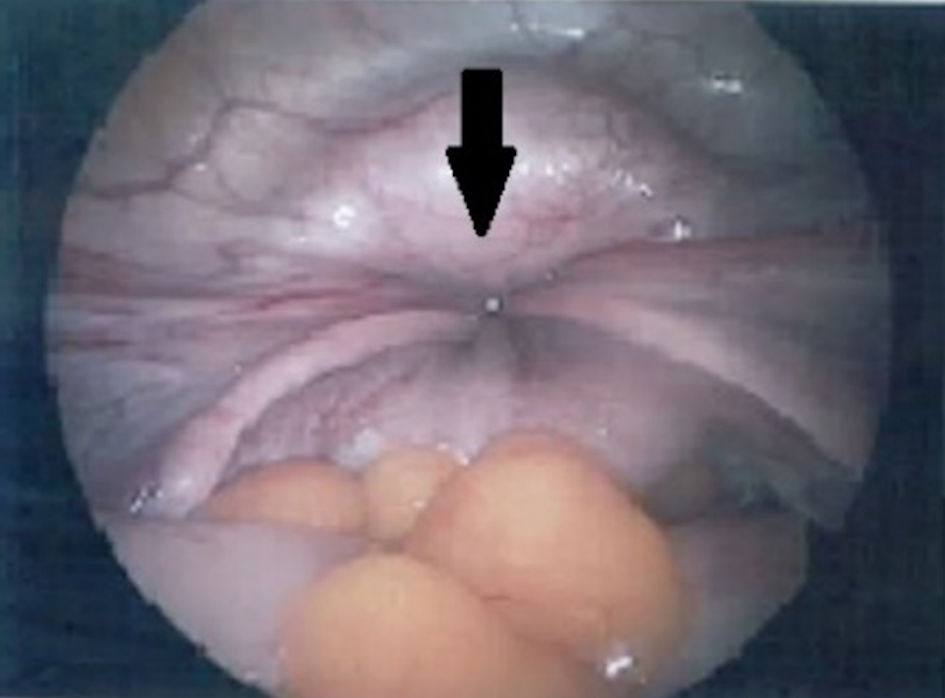 Click for large image | Figure 4. Image from diagnostic laparoscopy with arrow indicating site of uterine inversion with bilateral tubes and ovarian ligaments pulled in. |
Reduction of uterine inversion was attempted with O’ Sullivan’s hydrostatic method with normal saline and a Silastic cup (Ogueh and Ayida technique - modified from O’ Sullivan’s method), but this was unsuccessful. Manual reduction was then attempted but this was also unsuccessful.
The operation was converted to a laparotomy and manual reduction was achieved with Huntington’s method. Hysteroscopy was repeated after successful reduction of the uterus and the cervix was identified. The stalk of the mass could be seen at the fundus.
Postoperatively, the patient had an uneventful recovery, and she was discharged on postoperative day 5.
Follow-up and outcomes
The resected mass was sent for histological examination. In the specimen which measured 65 × 45 × 30 mm, there were polypoidal pieces of endometrial tissue containing atypical and closely packed endometrial glands. The endometrial glands were lined by highly atypical columnar epithelium with irregular nuclear membranes, conspicuous nucleoli, and hyperchromatic nuclei. There were also scattered bizarre cells with enlarged hyperchromatic nuclei present. In some areas, there were solid islands of atypical cells with vesicular nuclei and abundant eosinophilic cytoplasm were present. There were also foci of vascular invasion identified. The atypical glands were positive for vimentin, epithelial membrane antigen (EMA), focally positive for p16 and p53 (wild type) but were negative for hepatocyte nuclear factor (HNF)-1beta and desmin. Based on the immunoprofile and morphology, the diagnosis was that of grade 3 endometrioid adenocarcinoma. Staging computed tomography (CT) scans of the thorax, abdomen, and pelvis showed no distant metastasis or systemic adenopathy (Figs. 5, 6).
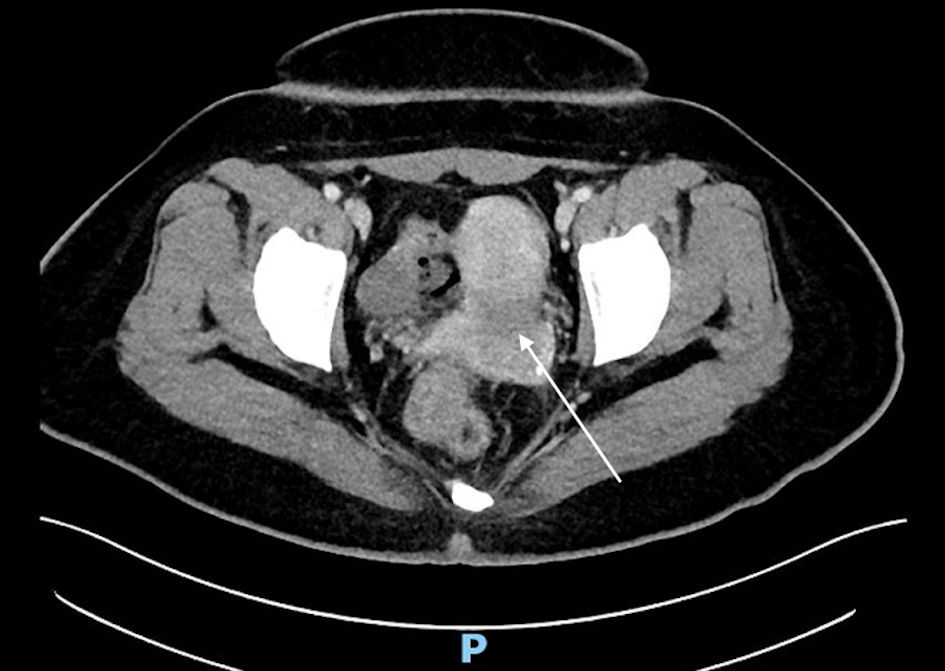 Click for large image | Figure 5. Computed tomography scan image (axial section) with white arrow indicating heterogenous hypodensity within the endometrial cavity. |
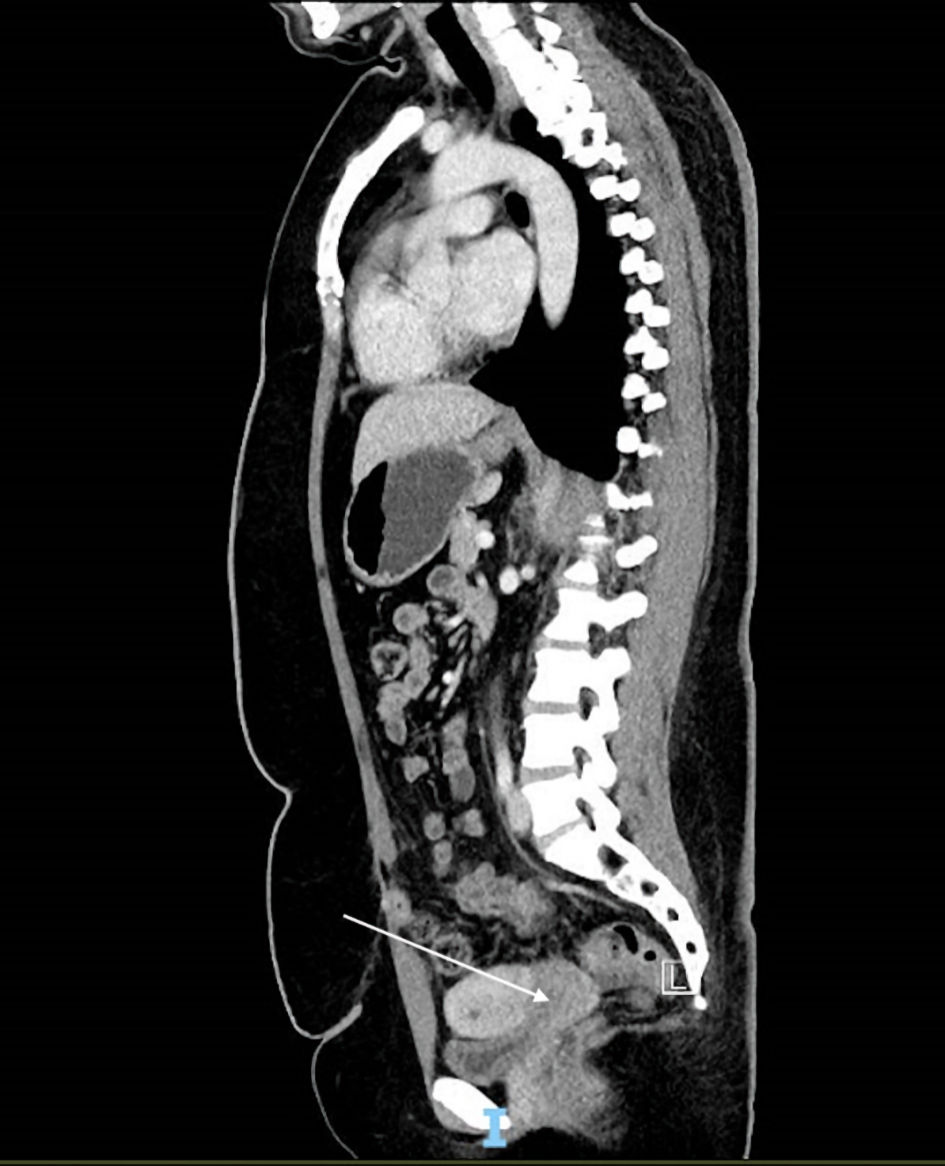 Click for large image | Figure 6. Computer tomography scan image (sagittal section) with white arrow indicating heterogenous hypodensity within the endometrial cavity which possibly extends to the endocervical canal. |
The patient then underwent completion total hysterectomy, bilateral salpingo-oophorectomy (THBSO), pelvic lymph node dissection, para-aortic node dissection, and adhesiolysis. Final histology revealed that there was no residual malignancy, and all lymph nodes were also negative for malignancy. The staging based on the International Federation of Gynecology and Obstetrics (FIGO) staging system was stage 1A.
Following discussion at a multidisciplinary tumor board, she received adjuvant brachytherapy a month following her second surgery.
At the time of the last follow-up visit, she remained well and was planned for a repeat follow-up visit in 6 months’ time.
| Discussion | ▴Top |
Non-puerperal uterine inversion occurs so rarely that there is no good estimate of its incidence [2]. It can be classified into four stages depending on the degree of inversion (Table 1): stage 1 being incomplete inversion with the uterine fundus remaining within the cavity; stage 2 being complete inversion of the uterine fundus through the fibromuscular ring of the cervix; stage 3 being total inversion with the fundus protruding through the vulva; and stage 4 with the vagina also involved and inverted through the vulva along with the uterus [8]. Although majority of the cases are secondary to benign causes, it is important to obtain a biopsy of the tumor before treatment. If it is a malignant tumor, definitive surgical management would require total hysterectomy and bilateral salpingo-oophorectomy which may be performed in the same setting as repositioning of the uterus. In this patient, as she was young, the possibility of the large mass being malignant was not discussed prior to the first surgery which she underwent. Therefore, it resulted in a second surgery that was required for her treatment.
 Click to view | Table 1. Stages of Uterine Inversion |
The mechanism by which uterine inversion occurs is thought to be due to a combination of factors, such as weakening of the uterine wall by the tumor, size of the tumor, thickness of the tumor pedicle, sudden extrusion of the tumor from the uterus, and uterine attachment site of the tumor [6, 9, 10].
In the case described above, there was sudden extrusion of the tumor and dilatation of the cervix due to oral misoprostol that had been administered in preparation for hysteroscopy, dilatation and curettage, and TCRM. The tumor had likely been present for a prolonged duration as the patient had been experiencing symptoms for a few years. This would have led to chronic weakening of the uterine wall. The tumor was also attached to the fundus. The presence of these multiple factors likely culminated in uterine inversion.
The diagnosis of uterine inversion is challenging and can only be made when there is a high index of suspicion. In the non-puerperal setting, patients commonly present with vaginal bleeding, vaginal mass, pelvic pain, abdominal discomfort, and anuria [6, 11]. On physical examination, a diagnosis of uterine inversion may be suspected with the presence of a mass in the vagina or at the introitus, unable to palpate the fundus properly and inability to visualize the cervix [6].
In this patient who was virgo intacta and never had sexual intercourse previously, routine speculum and bimanual vaginal examination were not performed at presentation in view of the anticipated discomfort which the patient would experience with such examination methods. This eventually led to a delayed diagnosis of a prolapsed fibroid polyp as the cause of the patient’s symptoms. In the assessment of patients who are virgo intacta, consideration may be given to the use of special specula such as the Huffman speculum, nasal speculum, or the use of a hysteroscope. If these equipment are not available, consent may be obtained from the patient to perform recto-abdominal bimanual palpation. In a relaxed and cooperative patient, information may be obtained regarding the shape and size of the uterus, the contents of the pouch of Douglas (POD), and even adnexal pathology if there are any masses present [12]. Imaging modalities in the form of ultrasound and MRI may also aid the diagnosis. If the patient does not consent to transvaginal ultrasound in view of her virgo intacta status, transabdominal, transperineal, or transrectal approaches may be undertaken. On ultrasound imaging, the uterine inversion will be seen as an indentation of the fundic area and a depressed longitudinal groove extending from the uterus to the center of the inverted portion [6]. On MRI, sagittal views may reveal a U-shaped uterine cavity and a thickened and inverted uterine fundus while axial views may show a “bull’s-eye” appearance [9, 13].
Management of non-puerperal uterine inversion involves adequate resuscitation on initial presentation as patients usually have heavy vaginal bleeding. After the diagnosis of uterine inversion is made, the patient should be brought to the operating theatre to attempt reduction of the inversion. Patients should be counseled on the option of hysterectomy especially if they are perimenopausal or menopausal. The different routes for attempted reduction (Table 2) - vaginal, laparoscopic, or laparotomy - should also be discussed. Patients should be warned that if final histology returns to be malignancy, a second surgery may be required. Conservative methods may be attempted first, such as manual reduction via Johnson’s maneuver (placing a hand inside the vagina and pushing the fundus along the long axis of the vagina towards the umbilicus) or hydrostatic method via O’ Sullivan’s method (infusing warm saline into the vagina, therefore creating a water seal with the operator’s hand and the vulva), although these methods are seldom successful [6, 14].
 Click to view | Table 2. Methods for Reduction of Uterine Inversion |
The surgical approaches available would be Huntington’s method (sequential clamping and traction on the round ligaments) or the Haultain procedure (incising the posterior surface of the uterus to bisect the constriction ring) which are performed abdominally and Spinelli (transecting the cervix anteriorly) or Kustner’s (transecting the cervix posteriorly) methods which are performed vaginally [6, 14].
The cause of the uterine inversion should also be treated. For benign tumors, these should be excised. As for malignant tumors, they should be managed accordingly with definitive surgical management after being appropriately staged.
This case presents the unique challenges of making the diagnosis of non-puerperal uterine inversion secondary to endometrial malignancy in a young patient who was virgo intacta. Her state of virgo intacta presented complexities in physical examination as routine methods of examination were not suitable, which added to the difficulty in making the diagnosis. Her young age also made malignancy lower down in the list of differentials when she was counseled for her first surgery, and unfortunately, this resulted in a second surgery being required to treat her condition.
Learning points
A high index of suspicion needs to be present in order to make the diagnosis of non-puerperal uterine inversion. In the assessment of the virgo intacta patient, challenges may be overcome if consent may be obtained for recto-abdominal bimanual palpation and the use of different approaches in performing ultrasonography of the pelvis. As malignant causes make up almost 15% of causes of non-puerperal uterine inversion, obtaining a histological diagnosis before definitive surgical management is undertaken might save patients the need to undergo repeat laparotomy if repositioning of the uterus and THBSO may be performed in a single operation. Therefore, adequate counseling of patients before surgery with discussion of possible outcomes including need for repeat surgery is crucial.
Acknowledgments
None to declare.
Financial Disclosure
None to declare.
Conflict of Interest
None to declare.
Informed Consent
Informed consent was obtained from the patient prior to the write-up of the case report with documentation on a consent form.
Author Contributions
The authors confirm contribution to the paper as follows: data collection: W. Wang; draft manuscript preparation: W. Wang and J. Wang. All authors approved the final version of the manuscript.
Data Availability
Any inquiries regarding supporting data availability of this study should be directed to the corresponding author.
Abbreviations
CT: computed tomography; THBSO: total hysterectomy, bilateral salpingo-oophorectomy; TCRP: transcervical resection of polyp; TCRM: transcervical resection of myoma; MRI: magnetic resonance imaging
| References | ▴Top |
- Das P. Inversion of the uterus. J Obstet Gynaecol Br Emp. 1940;47:525-548.
doi - Gowri V. Uterine inversion and corpus malignancies: a historical review. Obstet Gynecol Surv. 2000;55(11):703-707.
doi pubmed - Lupovitch A, England ER, Chen R. Non-puerperal uterine inversion in association with uterine sarcoma: case report in a 26-year-old and review of the literature. Gynecol Oncol. 2005;97(3):938-941.
doi pubmed - Mwinyoglee J, Simelela N, Marivate M. Non-puerperal uterine inversions. A two case report and review of literature. Cent Afr J Med. 1997;43(9):268-271.
- da Silva BB, Dos Santos AR, Bosco Parentes-Vieira J, Lopes-Costa PV, Pires CG. Embryonal rhabdomyosarcoma of the uterus associated with uterine inversion in an adolescent: a case report and published work review. J Obstet Gynaecol Res. 2008;34(4 Pt 2):735-738.
doi pubmed - Buyukkurt S, Vardar MA, Zeren H, Ozgunen FT. Non-puerperal inversion of the uterus caused by leiomyosarcoma: a case report and clinical management. J Obstet Gynaecol Res. 2007;33(3):402-406.
doi pubmed - Herath RP, Patabendige M, Rashid M, Wijesinghe PS. Nonpuerperal Uterine Inversion: What the Gynaecologists Need to Know? Obstet Gynecol Int. 2020;2020:8625186.
doi pubmed - Skinner GN, Louden KA. Non-puerperal uterine inversion associated with an atypical leiomyoma. Aust N Z J Obstet Gynaecol. 2001;41(1):100-101.
doi pubmed - Moodley M, Moodley J. Non-puerperal uterine inversion in association with uterine sarcoma: clinical management. Int J Gynecol Cancer. 2003;13(2):244-245.
doi pubmed - Takano K, Ichikawa Y, Tsunoda H, Nishida M. Uterine inversion caused by uterine sarcoma: a case report. Jpn J Clin Oncol. 2001;31(1):39-42.
doi pubmed - Gomez-Lobo V, Burch W, Khanna PC. Nonpuerperal uterine inversion associated with an immature teratoma of the uterus in an adolescent. Obstet Gynecol. 2007;110(2 Pt 2):491-493.
doi pubmed - Kruger TF, Botha MH. Clinical gynaecology, 3rd ed. Cape Town, Juta, South Africa. 2007. p. 16.
- Lewin JS, Bryan PJ. MR imaging of uterine inversion. J Comput Assist Tomogr. 1989;13(2):357-359.
doi pubmed - Rocconi R, Huh WK, Chiang S. Postmenopausal uterine inversion associated with endometrial polyps. Obstet Gynecol. 2003;102(3):521-523.
doi pubmed
This article is distributed under the terms of the Creative Commons Attribution Non-Commercial 4.0 International License, which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Journal of Medical Cases is published by Elmer Press Inc.


