| Journal of Medical Cases, ISSN 1923-4155 print, 1923-4163 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Med Cases and Elmer Press Inc |
| Journal website https://www.journalmc.org |
Case Report
Volume 14, Number 3, March 2023, pages 105-110
Subarachnoid Hemorrhage From Cavernous Malformation Masquerading as Myocardial Infarction
Mohammed S. Abdallaa, b, Ben Carlton Smitha, Allison Kirchnera, Maria Abu Nseira, Mousab Mokhtara, Ahmed Abdulrahmana, Eltaib Saada
aDepartment of Internal Medicine, Ascension Saint Francis Hospital, Evanston, IL, USA
bCorresponding Author: Mohammed S. Abdalla, Department of Internal Medicine, Ascension Saint Francis Hospital, Evanston, IL 60202, USA
Manuscript submitted February 21, 2023, accepted March 28, 2023, published online March 31, 2023
Short title: SAH Masquerading as Myocardial Infarction
doi: https://doi.org/10.14740/jmc4064
| Abstract | ▴Top |
Intracranial hemorrhage, including subarachnoid hemorrhage (SAH), is associated with many cardiac effects, including cardiac rhythm abnormalities, ischemic electrocardiographic (ECG) changes, elevated cardiac troponin levels, and regional wall motion abnormalities on echocardiogram. About 40% of patients with SAH demonstrate increased serum markers for myocardial necrosis. Approximately 10% of patients with SAH demonstrate left ventricular (LV) wall motion abnormalities; a subset of these patients will have irreversible myocardial damage, but most regain LV function in several weeks. Cardiac effects of SAH are thought to be a result of an imbalance of the autonomic nervous system with resultant increased catecholamine effect on the myocardial cells rather than due to preexisting coronary artery disease. These cardiovascular complications carry a prognostic significance in patients with SAH and can also be misdiagnosed as primary cardiac problems and delay the diagnosis of SAH. Herein, we present a case of a 68-year-old female who presented to the emergency department with acute onset of upper back and neck pain. She was initially misdiagnosed with myocardial infarction in view of the ischemic changes in the ECG and elevated cardiac troponins. She was started on antiplatelets and anticoagulation but was later found to have a negative coronary angiography and was diagnosed with SAH via a computed tomography (CT) scan. Intracranial hemorrhage can be associated with elevated cardiac enzymes and ECG changes and can sometimes masquerade as an acute coronary syndrome (ACS). A careful history and examination and a high index of clinical suspicion are pivotal in such cases since early diagnosis significantly impacts prognosis and prevents the inadvertent use of antiplatelets and anticoagulation, which can be detrimental if used in such cases.
Keywords: Intracranial hemorrhage; Subarachnoid hemorrhage; Cardiac enzymes; Elevated troponin; ECG changes; Myocardial infarction
| Introduction | ▴Top |
Subarachnoid hemorrhage (SAH) is a common cause of stroke in adults accounting for about 10% of all stroke cases. The condition typically presents with severe headaches, often described as a “thunderclap” like, and can be associated with altered mental status and focal neurological deficits [1]. In some patients, however, these typical clinical manifestations may not be present, and the condition may present more subtly, leading to delays in diagnosis and timely institution of appropriate treatment, the clinical consequences of which can be devastating [2, 3].
SAH can sometimes present with symptoms seen with acute coronary syndrome (ACS), such as upper back, shoulder, or neck pains, and although atypical, these symptoms can often be confused for ACS because of the commonly associated electrocardiographic (ECG) changes and elevation in cardiac troponins [4-6]. Herein we present a patient who presented to the emergency department (ED) with upper back and neck pain, diagnosed initially as non-ST segment elevation myocardial infarction (NSTEMI) but was eventually found to have occipital SAH after extensive inpatient workup.
| Case Report | ▴Top |
Investigations
The patient is a 68-year-old female with a past medical history of hypertension and migraine headaches who presented to the ED with neck, left shoulder, and upper back pain for 1 day. The pain started while the patient was resting at home. The pain improved partially with paracetamol tablets. Later in the day, the patient stated she tried to pick up an item from the ground when her severe neck/back pain returned and has since persisted. Her neck pain was not associated with chest pain, diaphoresis, nausea, vomiting, dizziness, lightheadedness, or limb weakness. She has not reported any recent falls or trauma. The patient initially presented to an urgent care center where ECG demonstrated ST segment depression, and the patient was advised to go to the ED for further evaluation. The patient noticed an improvement in neck pain upon arrival at the ED. However, the pain was somehow worse with movement; she did not report headaches, focal weakness, paresthesia, or bowel or urinary symptoms.
Diagnosis
Given the patient’s clinical presentation and ECG findings, ACS was suspected. ECG in the ED showed ST depression in leads V3-V6 (Fig. 1a), and high-sensitivity troponin level was significantly elevated to > 4,800 (0 - 20 pg/mL). The patient was started on antiplatelet therapy and anticoagulation with heparin. Repeat ECG on the next day showed improvement of the ST deviation noticed on the first day (Fig. 1b). Echocardiogram showed normal left ventricular wall thickness and normal ejection fraction of 60-65% without any regional wall motion abnormalities or other significant structural abnormalities. Cardiology was consulted, and the patient underwent urgent coronary catheterization on the second day of hospitalization; however, coronary angiography (Fig. 2a, b) did not reveal any significant coronary artery disease. Given the level of troponin rise, persistent upper back pain, and negative coronary angiography, a workup for aortic dissection was done with computed tomography (CT) angiography (Fig. 2d), which was again negative. Additionally, CT pulmonary angiography (Fig. 2c) was negative for pulmonary embolism. On the third day of hospitalization, the patient reported headaches and photophobia. The headaches were initially attributed to migraines which the patient stated typically worsen with stress. However, her headaches worsened, and she reported worsening neck pain and nausea. Her physical examination remained unremarkable throughout this period without any focal neurological deficits or alteration in her mental status. A head CT (Fig. 3a) was obtained at this point, revealing left occipital SAH with mild cerebral edema and minimal midline shift.
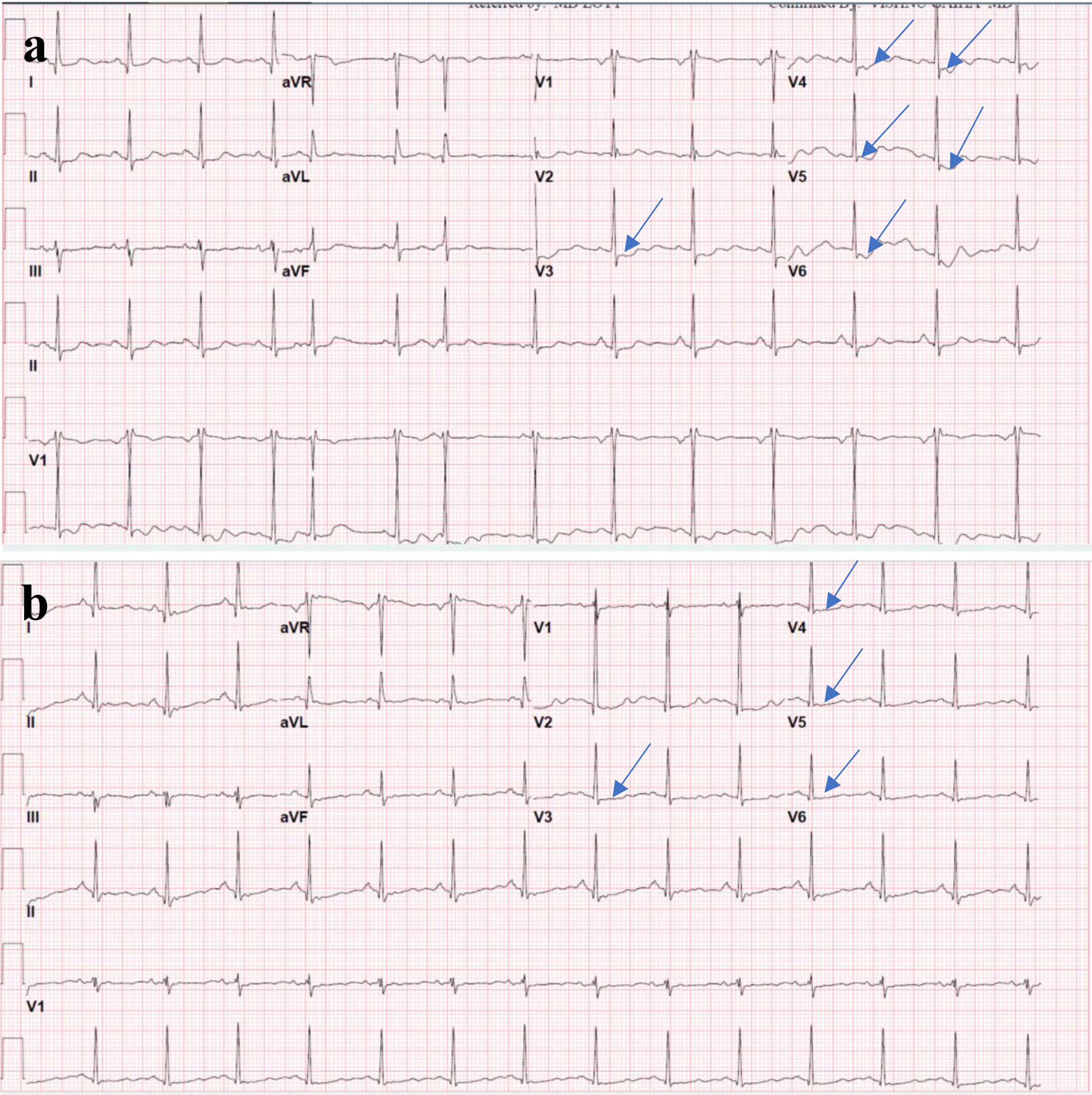 Click for large image | Figure 1. (a) ECG on presentation showing ST segment depression (arrows) in the anterolateral leads (V3-V6). (b) Repeat ECG on the next day showing improvement in the ST segment deviations noticed on the initial ECG. ECG: electrocardiogram. |
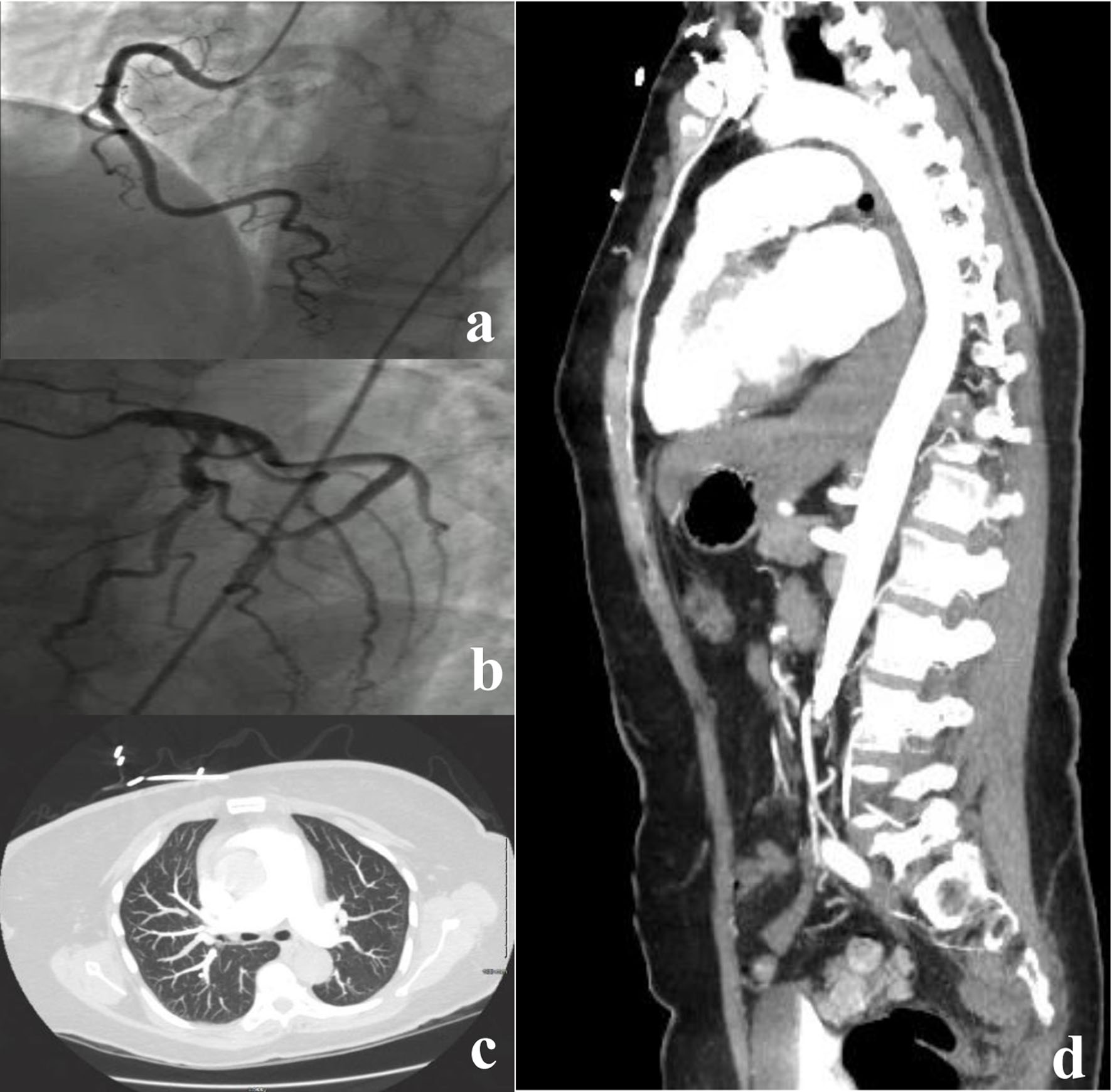 Click for large image | Figure 2. (a, b) Negative coronary angiography. (c) A single axial CT angiogram of the chest without a pulmonary artery filling defect, negative for pulmonary embolus. (d) A sagittal slice of a CT aortogram negative for aortic dissection. CT: computed tomography. |
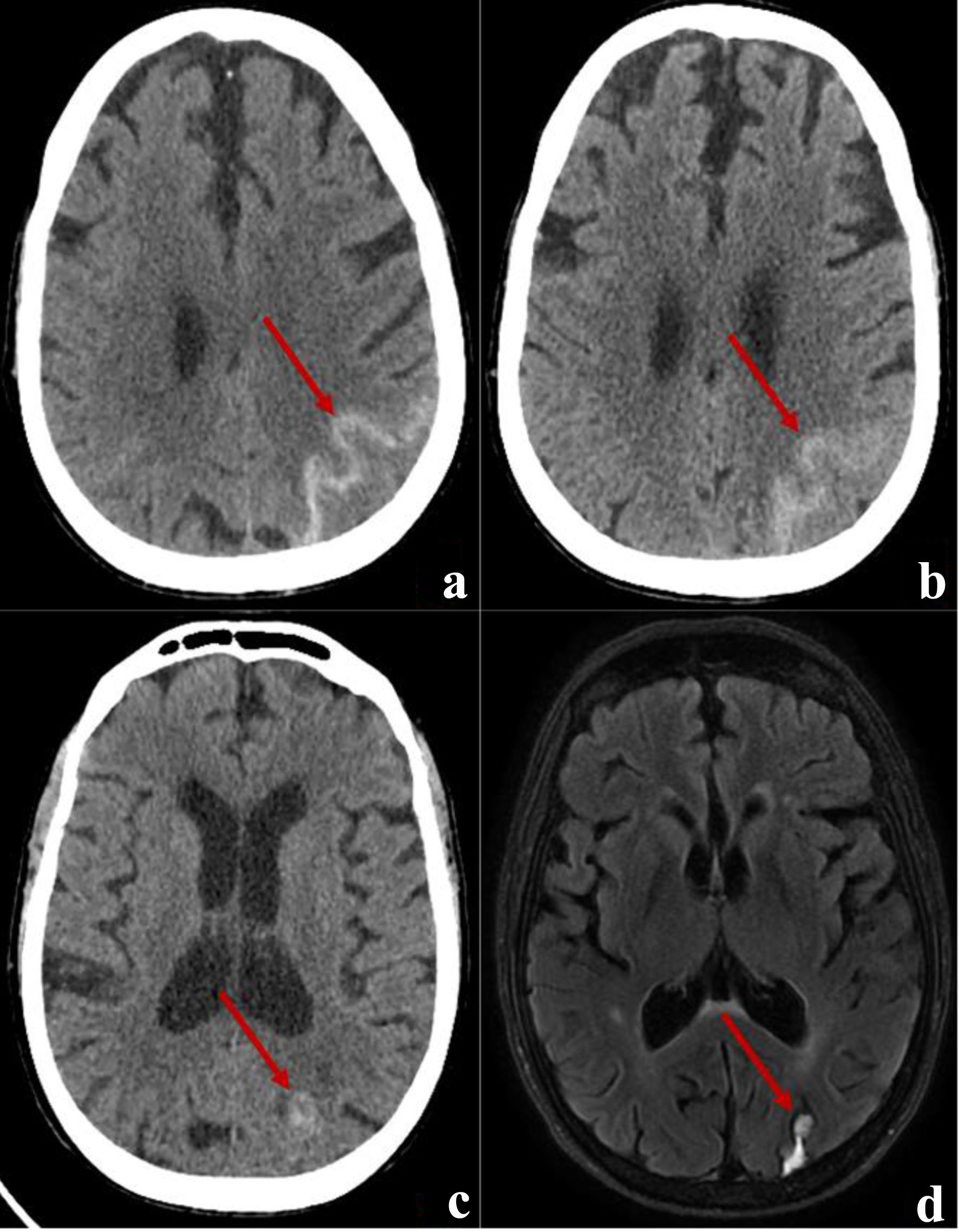 Click for large image | Figure 3. (a) An axial cut of a brain CT demonstrating acute SAH (arrow) in the left occipital lobe. (b) An axial cut of a CT brain demonstrating resolving SAH (arrow) on discharge. (c) The outpatient follow-up head scan at 3 weeks showing a resolved SAH and a focal hyperdensity (arrow) which raised suspicion for arteriovenous malformation. (d) A follow-up MRI 5 weeks after discharge showing a sub-centimeter cavernoma (arrow) adjacent to resolving SAH blood products. CT: computed tomography; SAH: subarachnoid hemorrhage; MRI: magnetic resonance imaging. |
Treatment
Antiplatelets and anticoagulation were held immediately upon diagnosis of SAH, and the patient was evaluated by a neurosurgeon. However, no neurosurgical intervention was recommended. The patient’s neurological status was observed in the intensive care unit (ICU) for 72 h. She remained neurologically intact, but her hospitalization was complicated by moderate hyponatremia due to the syndrome of inappropriate antidiuretic hormone (SIADH) in the context of her intracranial hemorrhage. The patient’s hyponatremia resolved with fluid restriction.
Follow-up and outcomes
After a week of hospitalization, the patient was discharged home in good condition without neurological deficits. A repeat head CT scan (Fig. 3b) showed a resolving SAH before discharge. At the follow-up in the neurosurgery clinic 3 weeks later, the patient had no neurological deficits, and the follow-up CT scan (Fig. 3c) showed a healing SAH with a suspicion of an underlying vascular malformation. Magnetic resonance imaging (MRI) of the brain (Fig. 3d) confirmed a diagnosis of cavernous malformation.
| Discussion | ▴Top |
Twenty percent of strokes in adults are due to intracranial hemorrhage, with SAH and intracerebral hemorrhage, each accounting for 10%. Most patients with spontaneous SAH have an underlying intracranial saccular aneurysm. However, about 20% of SAH cases are not due to a ruptured intracranial aneurysm. These are caused by arteriovenous malformations/fistulae, intracranial arterial dissection, amyloid angiopathies, vasculitides, bleeding disorders, and illicit drug use. Among these non-aneurysmal etiologies are cavernous malformations (CMs). CMs are very rare, with an incidence of 0.15 to 0.56 per 100,000 in various populations [7]. The incidence is equal in males and females, with a mean age of 30 to 40. However, females are more likely to present with intracranial hemorrhage and neurological deficits [8-10].
The typical clinical presentation of SAH is severe headache, which is sufficiently characteristic that an immediate non-contrast head CT scan should be performed as part of the evaluation of such patients, even those who are alert and neurologically intact at the time of initial presentation [11, 12]. In addition to headache, other common symptoms of SAH include transient loss of consciousness, neck pain or stiffness, and vomiting [13]. In one case series, these were present in 9%, 75%, and 61% of patients, respectively [14]. Meningismus, often accompanied by lower back pain, may develop several hours after the bleed due to aseptic meningitis caused by the breakdown of blood cells within the cerebrospinal fluid (CSF) [15]. Head CT scan without contrast is the initial diagnostic test of choice for SAH [12]. A lumbar puncture should be performed if the head CT is negative [12].
Cardiovascular effects of intracranial hemorrhage are well known and may have some prognostic significance, but they can also present diagnostic challenges in a minority of patients with intracranial hemorrhage. Cardiovascular effects of intracranial hemorrhage were first described in 1903 by Cushing, who noted elevations in blood pressure and cardiac rhythm abnormalities in such patients [16]. In 1947, Byer et al described ECG changes in a patient with SAH [17]. ECG changes are expected in almost all patients with SAH [4], and up to 10% are noted to have a potentially lethal arrhythmia, such as ventricular tachycardia and ventricular fibrillation, which may account for some of the mortality in patients with SAH. In addition to ECG changes, cardiac troponin I (cTnI) elevations have also been described in case series of patients with SAH [5, 6]. In these reports, cTnI elevations correlated with the severity of the neurological injury and cardiovascular complications, including pulmonary edema, LV dysfunction, and hypotension requiring vasopressors. In addition, some metanalyses correlated elevated cardiac troponins with the severity of the stroke, poor neurological outcome, longer ICU stays, and death [18, 19]. One meta-analysis showed that peak cTnI levels were independent predictors of death or severe disability at discharge. However, this association was insignificant at follow-up in 3 months [6]. These findings suggest that cTnI should be measured in all patients with SAH, especially those with ECG or clinical signs of potential cardiovascular dysfunction, and support the concept that cardiovascular dysfunction in these patients may contribute directly to poor outcomes. Although ECG changes are seen in almost all patients with SAH, and cardiac troponin elevations correlate with the severity of SAH as mentioned above, our patient has a significant elevation of troponins out of proportion to the mild SAH.
Although the above-mentioned cardiovascular effects carry important prognostic significance, they may also present diagnostic challenges in some patients with SAH. Several case reports suggested that some SAH patients can be misdiagnosed with ACS upon initial presentation [20-23]. Such patients are more likely to present with clinical features that were not specific enough to suggest SAH as a potential diagnosis and have features suggestive of cardiac origin for their symptoms, including ECG changes, high cardiac troponins, and regional wall motion abnormalities on echocardiogram. Our patient was initially misdiagnosed with NSTEMI in the context of her atypical presentation with neck, shoulder, and upper back pain with ECG changes and significantly elevated cardiac troponins, and the absence of headache at the initial presentation. It was not until the patient developed headaches associated with nausea and vomiting on the third hospital day that a head CT scan was performed, which confirmed SAH. Fortunately, her recovery was uneventful, and she was discharged home without neurological deficits.
Misdiagnosis and delayed diagnosis of SAH are common and can result in delays in starting appropriate treatment and worse clinical outcomes [2, 3]. The most common cause of misdiagnosis is the misconception that patients with SAH should always appear sick, have altered mental status, or have focal neurological deficits when in fact, about 40% of patients lack these clinical features [24]. This misconception is a common reason not to perform CT scans on such patients. A systematic review conducted in 2017 identified three studies published from 1996 to 2007 in ED populations with a pooled misdiagnosis rate of 7% [25]. In that systematic review, initial misdiagnosis was independently associated with small SAH volume, normal mental status at presentation, and right-sided aneurysm location in patients with aneurysmal SAH [2]. Failure to order a head CT scan at the initial clinical encounter was the most common error, accounting for 73% of the misdiagnoses. Patients with intact mental status at initial presentation (45%) had the highest misdiagnosis rate, which was associated with a fourfold increase in mortality risk at 12 months and increased morbidity among survivors.
Conclusion
SAH is a common emergency with high mortality and morbidity. Misdiagnosis and delayed SAH diagnosis are common and associated with poor clinical outcomes. The typical clinical features of SAH include severe headaches, often described as thunderclap-like, altered mental status, and often focal neurological deficits. However, some cases can present subtly or present atypically with symptoms suggestive of other alternative diagnoses such as ACS. These atypical presentations often lead to delay in diagnosis or misdiagnosis and can have profound clinical consequences, especially if anticoagulation and antiplatelets were used for an incorrectly diagnosed ACS.
Learning points
The most common cause of misdiagnosis of SAH is the misconception that patients with SAH should always appear sick, have altered mental status, or have focal neurological deficits. Cardiac effects of SAH, including ECG changes, elevated cardiac troponins, and regional wall motion abnormalities, are common. These cardiac effects have prognostic significance and can be a source of diagnostic confusion in patients with SAH who do not present with the typical severe thunderclap-type headaches. Initiation of antiplatelet therapy and anticoagulation in such circumstances can have serious consequences. Clinicians should be aware of the misconceptions surrounding the clinical presentation of SAH and know about the cardiac effects of intracranial hemorrhage to avoid misdiagnosis and delayed diagnosis in patients who do not have the typical clinical manifestations of SAH.
Acknowledgments
The authors would like to thank the Department of Radiology for providing the images.
Financial Disclosure
No funding was received for this case report.
Conflict of Interest
There is no conflict of interest in this case report from all authors.
Informed Consent
The patient provided consent for publication.
Author Contributions
MAN and AK Contributed to conceptualizing and writing the first manuscript. BCS prepared the case presentation and relevant imaging. MAbu Reviewed the case presentation. MM, AA and ES performed the critical review and editing of the final draft. All authors agreed to the final draft.
Data Availability
Any inquiries regarding supporting data availability of this the study should be directed to the corresponding author.
| References | ▴Top |
- Edlow JA, Caplan LR. Avoiding pitfalls in the diagnosis of subarachnoid hemorrhage. N Engl J Med. 2000;342(1):29-36.
doi - Kowalski RG, Claassen J, Kreiter KT, Bates JE, Ostapkovich ND, Connolly ES, Mayer SA. Initial misdiagnosis and outcome after subarachnoid hemorrhage. JAMA. 2004;291(7):866-869.
doi - Ois A, Vivas E, Figueras-Aguirre G, Guimaraens L, Cuadrado-Godia E, Avellaneda C, Bertran-Recasens B, et al. Misdiagnosis Worsens Prognosis in Subarachnoid Hemorrhage With Good Hunt and Hess Score. Stroke. 2019;50(11):3072-3076.
doi - Davis TP, Alexander J, Lesch M. Electrocardiographic changes associated with acute cerebrovascular disease: a clinical review. Prog Cardiovasc Dis. 1993;36(3):245-260.
doi - Tung P, Kopelnik A, Banki N, Ong K, Ko N, Lawton MT, Gress D, et al. Predictors of neurocardiogenic injury after subarachnoid hemorrhage. Stroke. 2004;35(2):548-551.
doi - Naidech AM, Kreiter KT, Janjua N, Ostapkovich ND, Parra A, Commichau C, Fitzsimmons BF, et al. Cardiac troponin elevation, cardiovascular morbidity, and outcome after subarachnoid hemorrhage. Circulation. 2005;112(18):2851-2856.
doi - Goldstein HE, Solomon RA. Epidemiology of cavernous malformations. Handb Clin Neurol. 2017;143:241-247.
doi - Del Curling O, Jr., Kelly DL, Jr., Elster AD, Craven TE. An analysis of the natural history of cavernous angiomas. J Neurosurg. 1991;75(5):702-708.
doi - Robinson JR, Awad IA, Little JR. Natural history of the cavernous angioma. J Neurosurg. 1991;75(5):709-714.
doi - Ene C, Kaul A, Kim L. Natural history of cerebral cavernous malformations. Handb Clin Neurol. 2017;143:227-232.
doi - Perry JJ, Stiell IG, Sivilotti ML, Bullard MJ, Hohl CM, Sutherland J, Emond M, et al. Clinical decision rules to rule out subarachnoid hemorrhage for acute headache. JAMA. 2013;310(12):1248-1255.
doi - Edlow JA. Managing patients with nontraumatic, severe, rapid-onset headache. Ann Emerg Med. 2018;71(3):400-408.
doi - Claassen J, Park S. Spontaneous subarachnoid haemorrhage. Lancet. 2022;400(10355):846-862.
doi pubmed pmc - Perry JJ, Sivilotti MLA, Sutherland J, Hohl CM, Emond M, Calder LA, Vaillancourt C, et al. Validation of the Ottawa Subarachnoid Hemorrhage Rule in patients with acute headache. CMAJ. 2017;189(45):E1379-E1385.
doi pubmed pmc - Schievink WI. Intracranial aneurysms. N Engl J Med. 1997;336(1):28-40.
doi - Cushing H. The blood pressure reaction of acute cerebral compression illustrated by cases of intracranial hemorrhage. Am J Med Sci. 1903;125:1017–1044. Crossref Google Scholar.
- Byer E, Ashman R, Toth LA. Electrocardiograms with large, upright T waves and long Q-T intervals. Am Heart J. 1947;33(6):796-806.
doi - Zhang L, Wang Z, Qi S. Cardiac troponin elevation and outcome after subarachnoid hemorrhage: a systematic review and meta-analysis. J Stroke Cerebrovasc Dis. 2015;24(10):2375-2384.
doi - Memar Montazerin S, Chi G, Marandi R, Najafi H, Shojaei F, Lee JJ, Marszalek J, et al. Evaluation of cardiac troponin and adverse outcomes after aneurysmal subarachnoid hemorrhage: a systematic review and meta-analysis. Neurocrit Care. 2022;36(2):650-661.
doi - Yusuf SW, Bhalla KS, Champion JC. Intracranial bleed mimicking acute myocardial infarction. Intern Med J. 2007;37(5):339-340.
doi - Lewandowski P. Subarachnoid haemorrhage imitating acute coronary syndrome as a cause of out-of-hospital cardiac arrest - case report. Anaesthesiol Intensive Ther. 2014;46(4):289-292.
doi - Heo WJ, Kang JH, Jeong WS, Jeong MY, Lee SH, Seo JY, Jo SW. Subarachnoid Hemorrhage Misdiagnosed as an Acute ST Elevation Myocardial Infarction. Korean Circ J. 2012;42(3):216-219.
doi pubmed pmc - Cheng TO. Subarachnoid hemorrhage mimicking acute myocardial infarction. Int J Cardiol. 2004;95(2-3):361-362.
doi - Edlow JA, Malek AM, Ogilvy CS. Aneurysmal subarachnoid hemorrhage: update for emergency physicians. J Emerg Med. 2008;34(3):237-251.
doi - Tarnutzer AA, Lee SH, Robinson KA, Wang Z, Edlow JA, Newman-Toker DE. ED misdiagnosis of cerebrovascular events in the era of modern neuroimaging: A meta-analysis. Neurology. 2017;88(15):1468-1477.
doi pubmed pmc
This article is distributed under the terms of the Creative Commons Attribution Non-Commercial 4.0 International License, which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Journal of Medical Cases is published by Elmer Press Inc.


