| Journal of Medical Cases, ISSN 1923-4155 print, 1923-4163 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Med Cases and Elmer Press Inc |
| Journal website https://www.journalmc.org |
Case Report
Volume 13, Number 11, November 2022, pages 551-556
Rare Case of Primary Adenoid Cystic Carcinoma of the Lung With Bilateral Kidney and Liver Metastasis
Talwinder Nagia, e, Rannah Dabirib, Yousra Gheitb, Abanoub Zakia, Willy Ganc, Alberto Torresa, Touqir Zahrad, Sachin Suled
aInternal Medicine Residency, Florida Atlantic University Charles E. Schmidt College of Medicine, Boca Raton, FL, USA
bFlorida Atlantic University Charles E. Schmidt College of Medicine, Boca Raton, FL, USA
cNeurology Residency, Florida Atlantic University Charles E. Schmidt College of Medicine, Boca Raton, FL, USA
dInternal Medicine, Florida Atlantic University Charles E. Schmidt College of Medicine, Boca Raton, FL, USA
eCorresponding Author: Talwinder Nagi, Internal Medicine Residency, Florida Atlantic University Charles E. Schmidt College of Medicine, Boca Raton, FL, USA
Manuscript submitted September 26, 2022, accepted November 14, 2022, published online November 27, 2022
Short title: Primary ACC of the Lung With Metastasis
doi: https://doi.org/10.14740/jmc4016
| Abstract | ▴Top |
Adenoid cystic carcinoma (ACC) is a rare epithelial tumor that is found in the salivary glands. It is typically slow-growing and follows an indolent course. While it can hematogenously spread to the lungs, distant metastases are rarely reported. Primary ACC in the lung is not common and makes up only 0.04-0.2% of all primary lung tumors. In addition, metastasis of the ACC to liver and bilateral kidneys is not common frequently documented. In this case report, we present a patient with unusual metastases of ACC, as well as non-specific symptoms that warrant discussion for ACC as potential differentials in the appropriate clinical setting.
Keywords: Adenoid cystic carcinoma; ACC metastasis; Lung ACC; CAP chemotherapy
| Introduction | ▴Top |
Adenoid cystic carcinoma (ACC) is a malignant neoplasm that typically arises in the salivary glands. It is a rare epithelial tumor that accounts for 10% of all salivary tumors and 1-2% of malignant tumors of the head and neck [1]. Notably, it may also arise in the breast, digestive tract or lungs. Instances in which ACC is classified as a primary lung tumor are rare, accounting for only 0.04-0.2% of all primary lung tumors [2]. Previously, primary ACC of the lungs was referred to as a benign glandular neoplasm. However today, it is regarded as a low-grade bronchial carcinoma, due to its origin from the bronchial glands, and is a slow-growing, low-grade malignancy.
Primary ACC of the lung presents differently than other primary lung malignancies. In terms of demographic characteristics, primary ACC of the lung tends to occur in the younger patient population, affecting males and females equally. Since it is a rare occurrence, most information describing the correlation of smoking to development of primary ACC of the lungs is collected through case studies. These studies suggest that smoking may not be associated with its etiology, unlike other lung cancers such as small cell or squamous. The severity of symptoms differs from patient to patient, but the most common presentations include cough, hemoptysis, and shortness of breath. As a result, many patients’ risk was misdiagnosed with asthma or bronchitis. Additionally, some patients remain asymptomatic and are diagnosed incidentally after completing imaging for another ailment [3].
Metastasis of primary ACC of the lung to distant sites such as the kidney and liver is relatively uncommon, and is only accounted for by a small number of cases published in literature. Hematoxylin-eosin staining remains the principal method for diagnosing ACC of the lung, and yet it can be misdiagnosed as carcinoid or adenocarcinoma in nature. As a result, immunohistochemistry is an important tool that helps guide histological diagnosis [3]. We present a case of a 51-year-old male who was diagnosed with primary ACC of the lung through hematoxylin-eosin staining after being admitted for complaints of weight loss and cough.
| Case Report | ▴Top |
Investigations
A 51-year-old male with a past medical history of gastroesophageal reflux disease presented to the emergency department on request of his primary care physician due to abnormal outpatient imaging findings on computed tomography (CT) scan. For the past 8 months, the patient has had symptoms of cough, dyspnea on exertion, lightheadedness, and significant weight loss. The cough is productive with clear-white sputum, occasionally having traces of bright red blood. Due to the worsening of dyspnea and cough upon exertion, the patient has difficulties carrying out daily tasks. The patient was prescribed an albuterol inhaler due to cough and shortness of breath. However, since the medication did not alleviate symptoms, the patient discontinued use of the inhaler. In the past 8 months, the patient endorsed about 20 pounds of unintentional weight loss. For the past year, the patient also reports feeling feverish, with profuse night sweats requiring constant change of clothing and bedsheets. He also reports lower back pain on both sides that has progressively worsened since the onset, 8 months ago. Within the last year, he has been treated for pneumonia and completed a course of antibiotics. Past surgical history is significant for a left leg open reduction and internal fixation and chest tube placement in 1998 due to a motor vehicle accident. He is a previous smoker of one pack every 2 days for 15 - 20 years and stopped 5 years prior. He used to drink alcohol every day for the past 10 years and decreased the frequency 5 years ago. The patient has worked in the automotive industry since the age of 15 and endorsed frequent exposure to multiple chemicals and gas. Otherwise, the patient denied headache, dizziness, chest pain, palpitations, abdominal pain, or changes in his urination or bowel movements.
Diagnosis
In the emergency department, the patient was hemodynamically stable, afebrile, body mass index of 19, and oxygen saturation of 98% on room air. He did not appear to be in any acute distress and did not have labored breathing. Physical exam was only significant for decreased breath sounds on auscultation of the full posterior right lung field. There were no crackles, wheezing, or rhonchi appreciated on anterior and posterior lung auscultation. Mild pain was endorsed on superficial palpation of the lower back bilaterally. Lab work showed a leukocytosis of 10,800 µL with no other significant results. A chest X-ray, presented as Figure 1, was completed and showed complete opacification of the right hemithorax with a rightward mediastinal shift, and mild irregularity of the right mainstem bronchus. Further evaluation with a CT of the chest with contrast was ordered, shown in Figures 2-4. This was significant for complete collapse of the right lung, an ill-defined hypoattenuation in the right upper lobe measuring 6.1 × 5.3 cm ascending into the right mainstem bronchus, concerning for neoplasm. There was a small right pleural effusion and an additional 1.3 cm nodule at the left lung base. Prominent right paratracheal lymph nodes, a large subcarinal lymph node measuring 2.4 × 1.4 cm, and scattered calcified mediastinal lymph nodes were also found. In the upper abdomen, a hypodense hepatic lesion measuring 5.2 × 3.8 cm with an additional 1.3 cm lesion was found in the superior right hepatic lobe. There were multiple bilateral renal hypodensities, one measuring 2.9 cm in the right anterior interpolar region and a 1.6 cm lesion in the left upper pole. Due to these concerning findings, the patient was admitted to the medical floor. Thoracic surgery, oncology, and pulmonology were consulted.
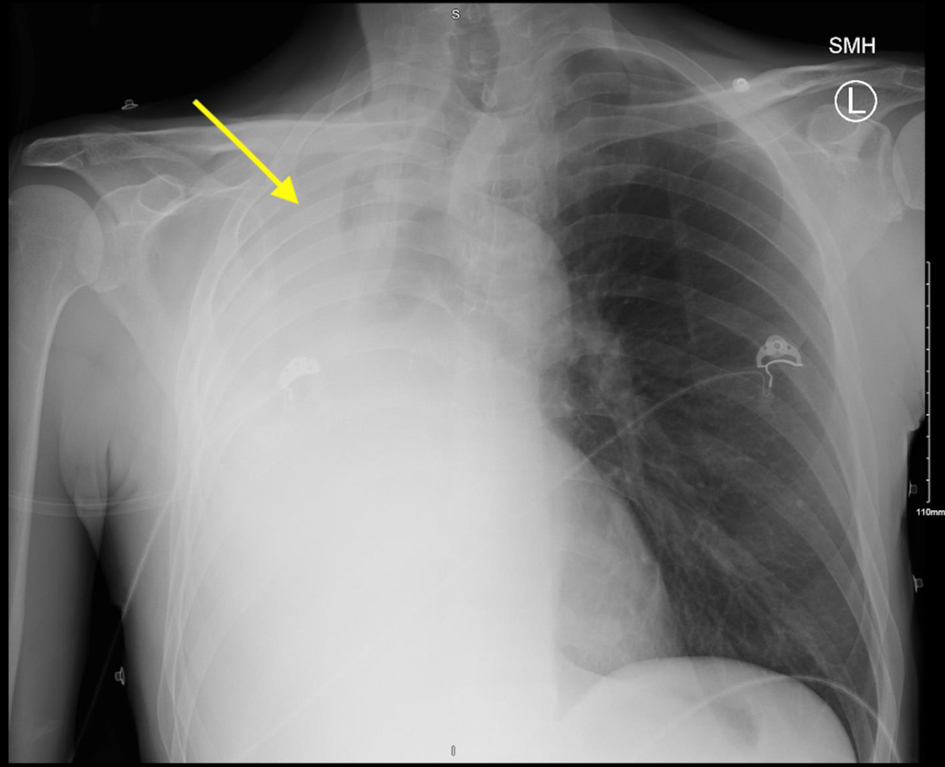 Click for large image | Figure 1. Chest X-ray with arrow depicting complete opacification of the right hemithorax with mediastinal shift to the right. Mild irregularity of the right mainstem bronchus. |
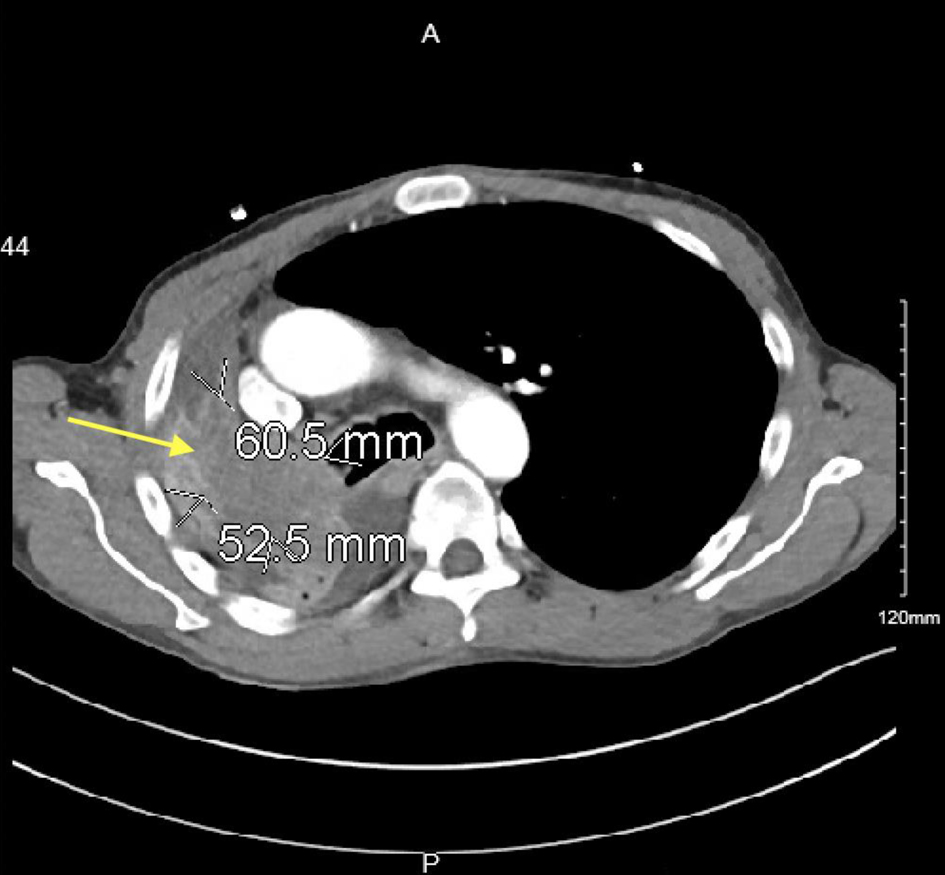 Click for large image | Figure 2. Contrast-enhanced CT image with yellow arrow depicting ill-defined hypoattenuation within the right upper lobe ascending into the right mainstem bronchus and bronchus intermedius measuring 6.1 × 5.3 cm. CT: computed tomography. |
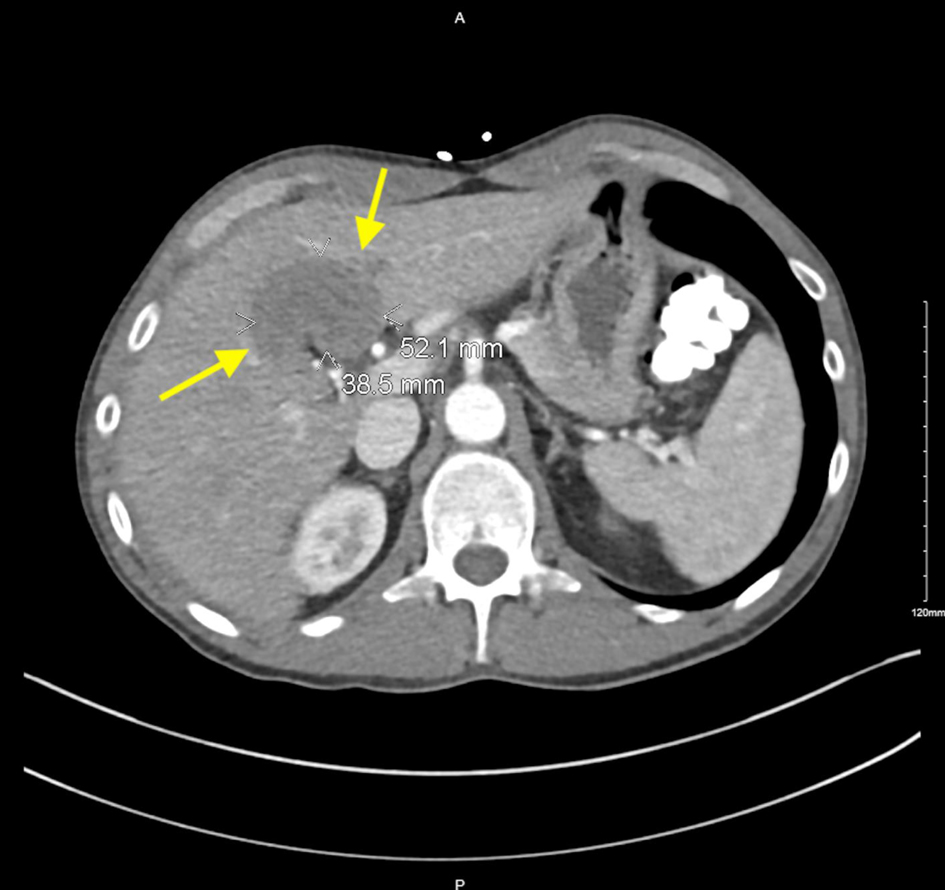 Click for large image | Figure 3. Contrast-enhanced CT image with yellow arrows pointing to hypodense lesion in the liver measuring 5.2 × 3.8 cm. CT: computed tomography. |
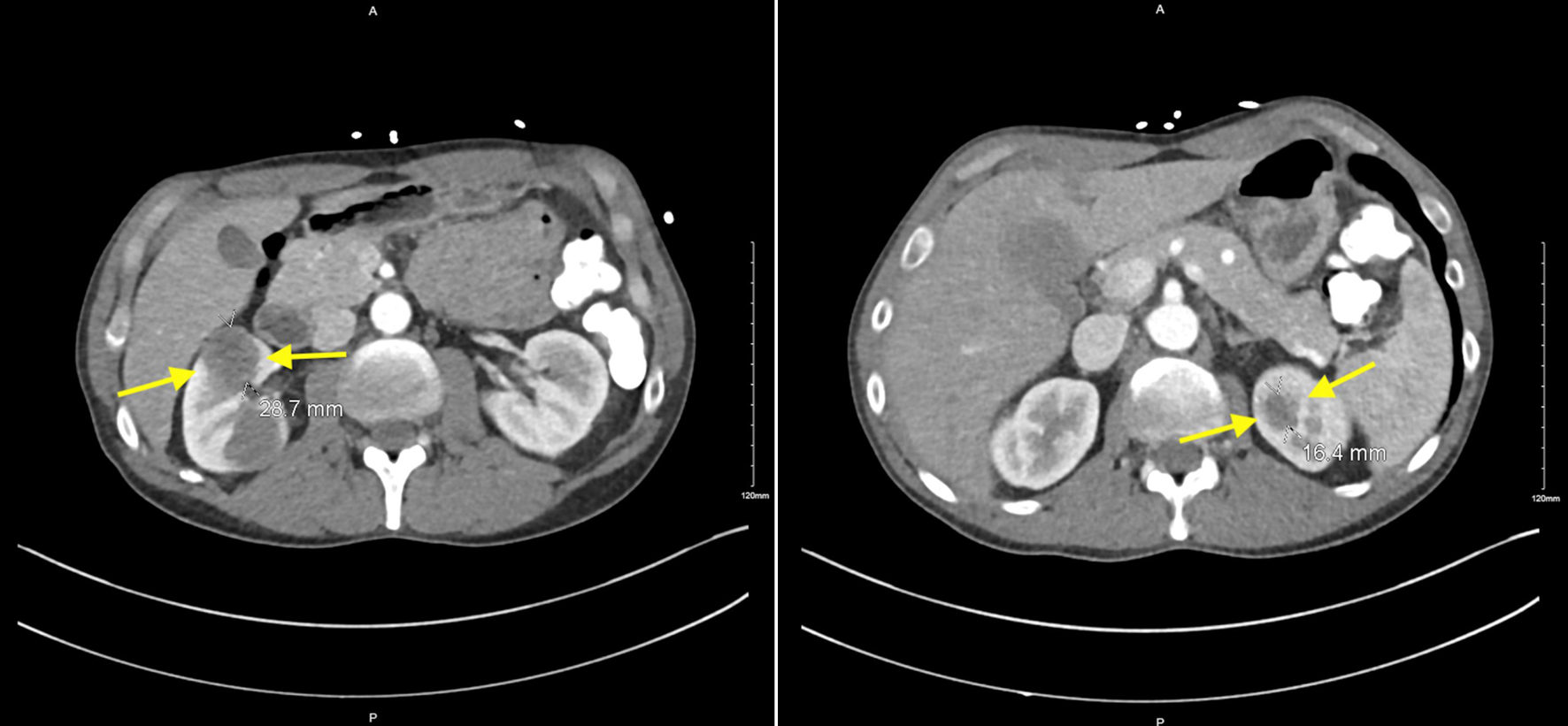 Click for large image | Figure 4. Contrast-enhanced CT imaging with yellow arrows depicting multiple bilateral renal hypodensities. Right anterior interpolar region measuring 2.9 cm and within the left upper pole measuring 1.6 cm. CT: computed tomography. |
A plan for a magnetic resonance imaging (MRI) of the brain with and without contrast was ordered due to the patient’s symptoms of lightheadedness and to rule out any brain involvement. A nuclear medicine (NM) bone scan was ordered to assess for any bone involvement, and a further plan for an ultrasound (US) thoracentesis and YAG laser bronchoscopy was made. The MRI of the brain was negative with no abnormal findings, and the NM bone scan showed no focal suspicious osseous uptake to suggest metastatic disease. The US thoracentesis was not completed due to an inadequate amount of fluid for a safe procedure, thus the patient was taken for the YAG laser bronchoscopy with biopsy. He was found to have a bulky tumor with complete obstruction of the right mainstem bronchus and biopsies were taken. Despite removing/lasering 3.5 cm of tumor, the airway still remained closed and a repeat rigid bronchoscopy with laser was planned. The patient tolerated repeat procedure well, and post-operative chest X-ray showed mild improved aeration in the right lung.
Pathology results from the biopsy of the mass were diagnostic for adenoid cystic carcinoma of the lung, depicted in Figures 5-7. Due to the liver and bilateral renal nodules noted on imaging, a CT-guided liver biopsy was planned to determine if this is metastatic disease. Biopsy results confirmed that the liver nodules were also adenoid cystic carcinoma, thus confirming metastasis.
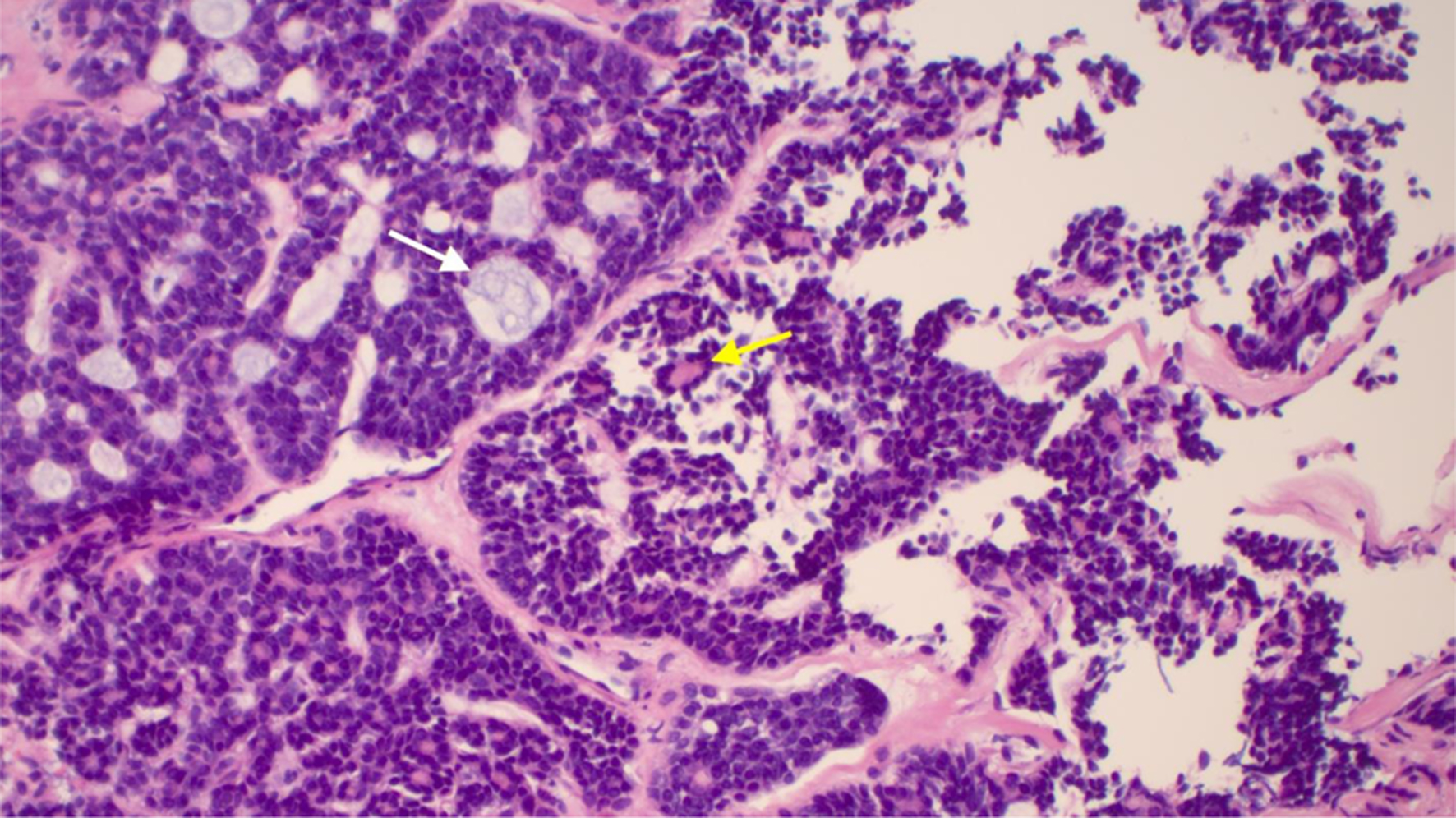 Click for large image | Figure 5. Right mainstem bronchus with adenoid cystic carcinoma in hematoxylin and eosin stain. White arrow depicts cribriform island containing basophilic mucin surrounded by myoepithelial cells. Yellow arrow depicts epithelial island with luminal cells. |
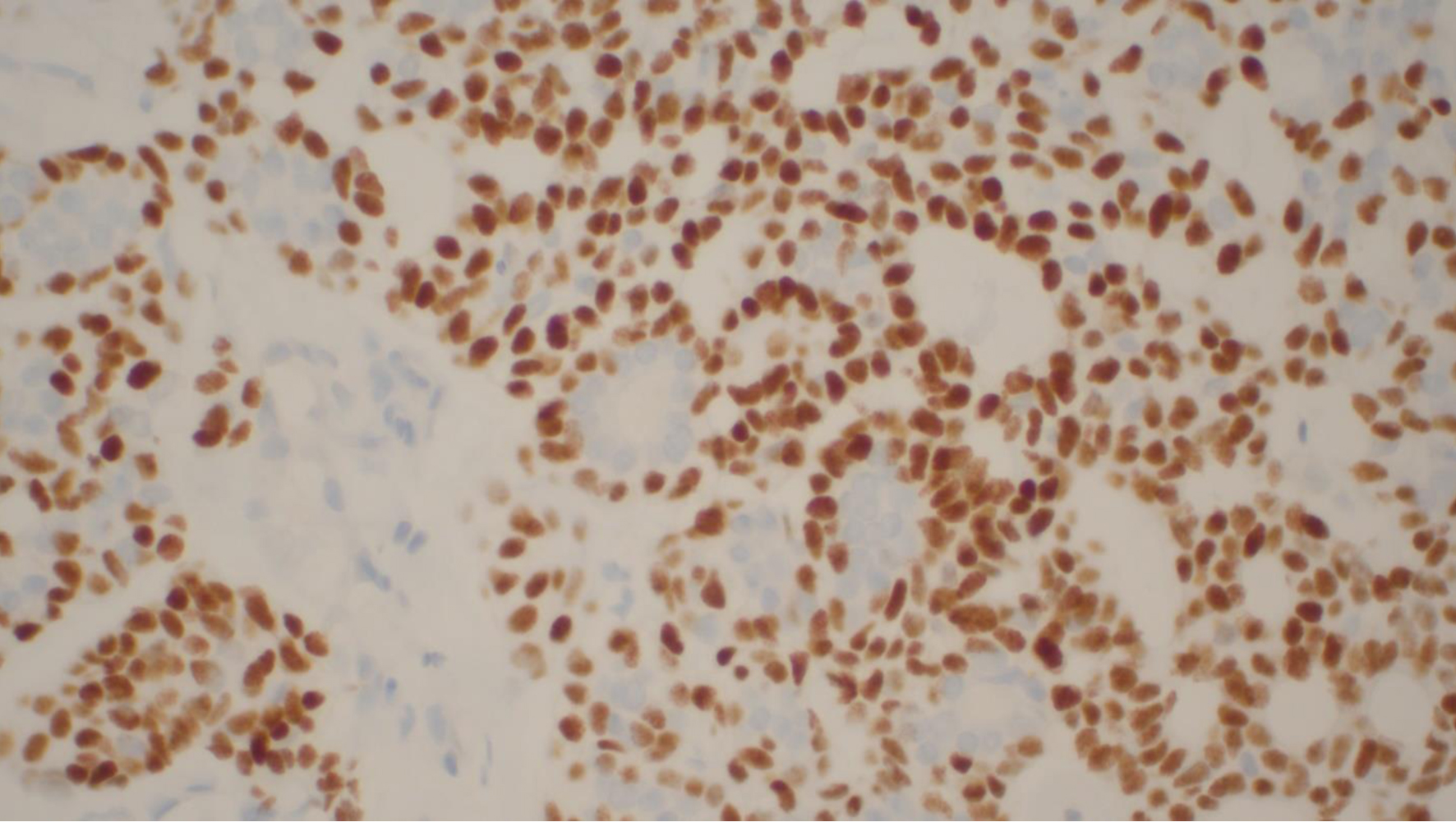 Click for large image | Figure 6. Adenoid cystic carcinoma with immunoperoxidase p40 staining nuclei of myoepithelial cells. Luminal epithelial cells are unstained. |
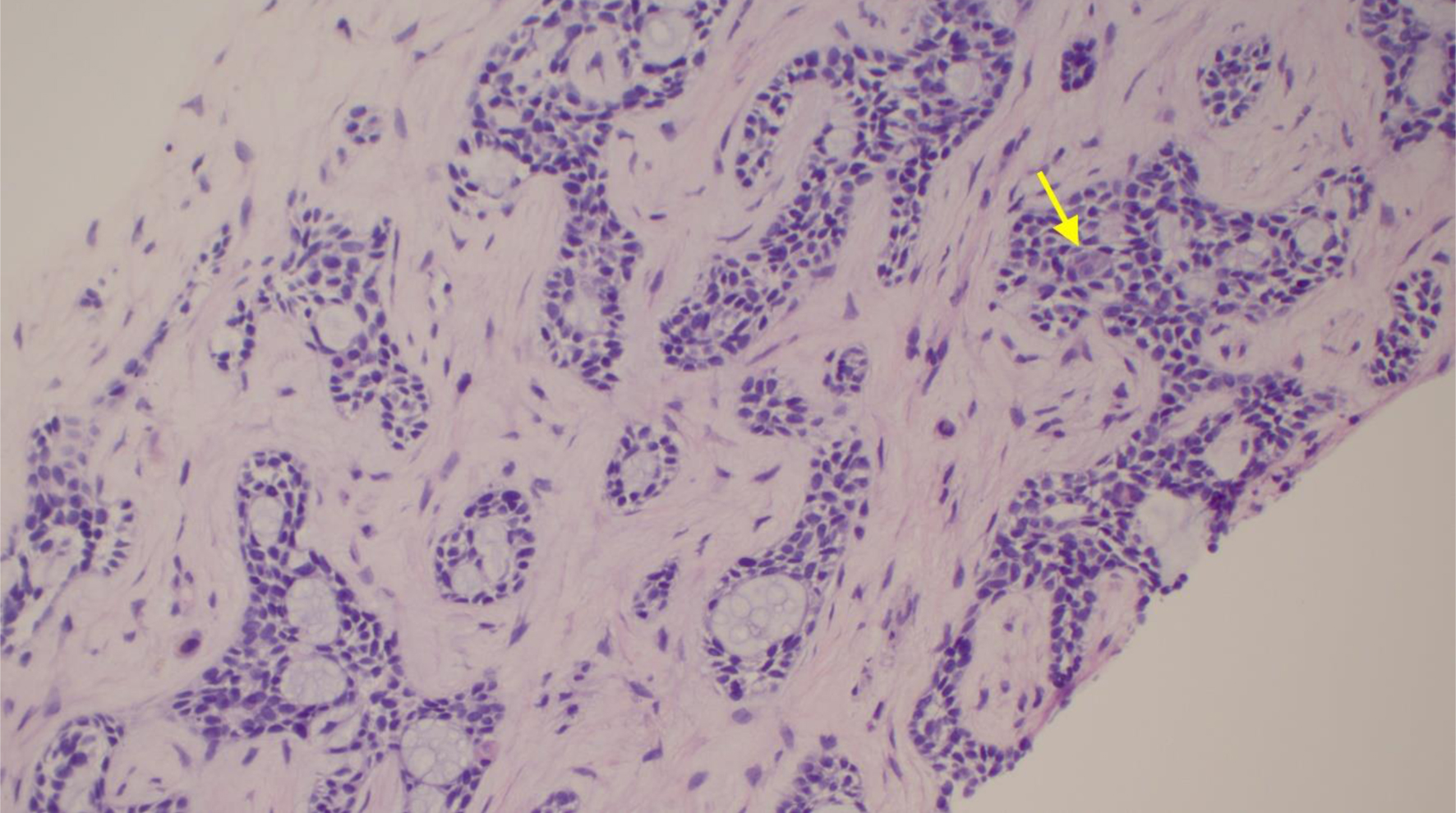 Click for large image | Figure 7. Section of liver core in hematoxylin and eosin stain showing identical morphology. Yellow arrow shows epithelial cells. Bluish mucin surrounding myoepithelial cells. |
Treatment and follow-up
With the exception of moderate cough and mild throat pain, the patient recovered well from bronchoscopy and was deemed stable for discharge from a medical standpoint. He followed up with a multimodal outpatient cancer clinic and due to progressive metastatic disease and worsening symptoms, received 5 days of radiation followed by four rounds of chemotherapy consisting of cyclophosphamide, adriamycin, and cisplatin (CAP).
| Discussion | ▴Top |
This case presents a patient with ACC, a salivary gland tumor that originated in the lung with subsequent metastasis to the liver and bilateral kidneys. ACC is an uncommon malignancy, representing approximately 1-2% of all cases of head and neck cancer and about 10% of all salivary neoplasms. ACC may also occur as a primary lung tumor, as presented in this case. It is considered malignant and arises centrally, specifically in the tracheobronchial glands in the airway submucosa. Previous studies reported that primary lung lesions were typically located in the trachea, main bronchus, or truncus intermedius, although some were found below the lobar bronchus or pulmonary parenchyma. Reportedly, it has a morphology that is similar to ACC that originates in the salivary glands [3].
In general, ACC is characterized by slow growth and perineural invasion, with distant dissemination being less likely. Although it tends to remain local, late recurrence after treatment is seen. For instance, one case study reported a 78-year-old male who presented with ACC that metastasized to his colon 19 years after the primary tumor was resected [4]. Due to the wide variability in site of metastasis and time following remission, serial abdominal imaging for recurrence may be beneficial.
Metastasis of ACC, particularly originating in the lung, is rare but is associated with poor prognosis and increased mortality [5]. In one study, only eight out of 34 participants with primary ACC of the lung had metastasis to other distant sites and were deemed as the non-operable group when considering treatment options. Of the eight, only one participant had metastasis to the kidney and two had metastasis to the liver such as presented in our case. Follow-up in this study showed that metastasis is associated with worse prognosis, to which patients eventually succumbed [3].
An additional case study reported a similar scenario as the case presented here. Namely, the patient was a 70-year-old male who had primary ACC of the lung and underwent treatment with right pneumonectomy and radiotherapy. Three years after initial presentation, he returned with symptoms of right flank pain and hematuria. MRI revealed solid renal lesions of which the largest was biopsied and found to be morphologically similar to his previous lung ACC, thus confirming renal metastasis. The patient was managed with a right nephrectomy and during follow-up 6 months later, the patient was alive and without disease [6]. To date, there have been only 11 instances in which this has occurred in the English literature.
Due to the indolent nature of ACC and the lack of effective treatment options, watchful waiting is typically recommended in asymptomatic individuals with stable disease. In individuals who are symptomatic, have progressing disease, or who are good surgical candidates, treatment is recommended. Surgical resection is the mainstay of treatment in primary ACC of the lung, in which the tumor is resectable. Particularly, a tracheobronchoplasty may be indicated since the tumor often arises centrally. Although it is common for resected margins to be positive due to widespread invasion along the airway, radiosensitivity is high and therefore postoperative radiotherapy may be a useful component in treatment.
Although the mainstay of medical therapy for advanced disease is still under debate, first-line treatment is often systemic chemotherapy. Current regimens include CAP as well as 5-fluorouracil and cisplatin. Overall effectiveness and tolerability of these regimens is unclear [7]. A phase II clinical trial looking at 22 individuals with advanced salivary gland carcinomas, of which 12 had ACC, showed an adjuvant CAP response rate of 27% [7]. With genomic studies of ACC identifying that the MYB-NFIB fusion gene is present in 70% of cases, focus is now shifting to use of immunotherapy [7]. Unfortunately, in our case this gene was not able to be assessed. Additionally, recent case studies have found that ACC of the lungs may show partial response to targeted therapies such as imatinib due to the expression of c-KIT [2]. Further studies should be done to determine efficacy of these targeted therapies.
This case highlights the rarity of primary lung ACC with distant metastasis and reviews the suggested workup for diagnosis along with treatment and surveillance. Although there is no definitive therapy recommended, chemotherapy and surgical resection remain the mainstay of treatment. Despite its infrequent diagnosis, ACC may not present with symptoms until the disease has progressed. Individuals affected may be treated for other conditions such as asthma or bronchitis since initial symptoms may be non-specific and overlooked. Thus, it is necessary to follow up with patients who have continued worsening symptoms and consider imaging for further evaluation.
Learning points
ACC is a rare tumor, accounting for only 1-2% of head and neck cancers, characterized by slow growth, local invasion, and delayed distant metastasis. Due to its indolent course, treatment is usually deferred while asymptomatic or in cases of stable disease, instead opting for regular monitoring. Typically, ACC metastases spread hematogenously to the lung or bones, but can also be seen in the liver, kidney, and colon. A retrospective study of 94 patients with ACC of the head and neck showed treatment failure in 67% of patients with 40% of those patients developing distant metastasis, 27% undergoing locoregional recurrence, and 33% developing both distant metastasis and locoregional recurrence [8]. Further, 95% of the individuals who developed pulmonary metastasis had no symptoms at the time of discovery [8]. Due to the unpredictable nature of ACC and the high incidence of recurrence, lifelong monitoring with imaging is prudent.
Acknowledgments
None to declare.
Financial Disclosure
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Conflict of Interest
None to declare.
Informed Consent
The manuscript has been de-identified. Informed consent was obtained from the patient.
Author Contributions
TN and RD wrote the case report, participated in the patient care, revised, and edited the case report, and approved the final version of the case report. YG wrote and edited the case report. AZ, WG, AT, TZ and SS revised and edited the case report and approved the final version of the case report.
Data Availability
The authors declare that data supporting the findings of this study are available within the article.
| References | ▴Top |
- Ammad Ud Din M, Shaikh H. Adenoid Cystic Cancer. In: StatPearls. Treasure Island (FL), 2022.
- Krifa M, Bdioui A, Lajmi Z, Missaoui N, Hmissa S, Mokni M. Primary adenoid cystic carcinoma of the lung: A case report and literature review. Heliyon. 2021;7(2):e06206.
doi pubmed - Moran CA, Suster S, Koss MN. Primary adenoid cystic carcinoma of the lung. A clinicopathologic and immunohistochemical study of 16 cases. Cancer. 1994;73(5):1390-1397.
doi - Mallikarjunappa SS, Jakate S, Cheng L. Colonic metastasis of adenoid cystic carcinoma 19 years after the primary tumor resection. Case Rep Pathol. 2021;2021:5511935.
doi pubmed - Jang S, Patel PN, Kimple RJ, McCulloch TM. Clinical outcomes and prognostic factors of adenoid cystic carcinoma of the head and neck. Anticancer Res. 2017;37(6):3045-3052.
doi - Junejo NN, Almusalam L, Alothman KI, Al Hussain TO. An unusual case report of pulmonary adenoid cystic carcinoma metastasis to the kidney. Case report and literature review. Urol Case Rep. 2019;27:100927.
doi pubmed - Chae YK, Chung SY, Davis AA, Carneiro BA, Chandra S, Kaplan J, Kalyan A, et al. Adenoid cystic carcinoma: current therapy and potential therapeutic advances based on genomic profiling. Oncotarget. 2015;6(35):37117-37134.
doi pubmed - Sung MW, Kim KH, Kim JW, Min YG, Seong WJ, Roh JL, Lee SJ, et al. Clinicopathologic predictors and impact of distant metastasis from adenoid cystic carcinoma of the head and neck. Arch Otolaryngol Head Neck Surg. 2003;129(11):1193-1197.
doi pubmed
This article is distributed under the terms of the Creative Commons Attribution Non-Commercial 4.0 International License, which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Journal of Medical Cases is published by Elmer Press Inc.


